Engineered for excellence, trusted by surgeons in 80+ countries.



Locking Plate Multi Axial for Distal Radius Fracture Orthopedic Surgery Implants

Titanium Coating Surgical Instrument Set Orthopedic Implant Cervical Interbody Fusion Peek Cage

Orthopedic Titanium Alloy Spine Surgical Instrument Set with CE/ISO/FDA

Ilizarov External Fixator -Arc Ring - Component of External Fixator


Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
As the global demand for spinal stability solutions increases, Anterior Cervical Plating (ACP) has emerged as the gold standard for stabilizing the cervical spine following discectomy and fusion (ACDF). Our facility stands at the forefront of this surgical revolution, combining decades of metallurgical expertise with advanced biomechanical research. We are not just manufacturers; we are the most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government.
Our commitment to E-E-A-T (Experience, Expertise, Authoritativeness, and Trustworthiness) is reflected in our massive global footprint, exporting to more than 80 countries worldwide. We pride ourselves on Superior Quality and Competitive Pricing, operating under a rigorous quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. This adherence to international protocols has made us the preferred partner for many reputed companies in Europe and the USA who regularly buy from us.
In an era of global supply chain volatility, our factory utilizes Industry 4.0 principles to ensure precision and continuity. Unlike many competitors, we don't compromise on quality in exchange for a cheaper price. We believe human life is invaluable, and our manufacturing process reflects this philosophy.
Our Quality Control starts from the raw material stage. This vertical integration makes it possible for us to control the quality of every component—from medical-grade titanium alloys to the final micro-polished surface. Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers.
 Automatic Slitting
Automatic Slitting CNC Lathe
CNC Lathe Laser Engraving
Laser Engraving Precision Grinding
Precision GrindingDriving the next generation of spinal implantology.
Development of ultra-thin plates (<1.5mm) to minimize post-operative dysphagia while maintaining structural integrity and fatigue resistance.
Advanced titanium plasma spray and hydroxyapatite coating technologies to accelerate osseointegration and reduce healing time.
Integration of digital torque sensors and navigational guides to assist surgeons in high-precision screw placement.
The future of Anterior Cervical Plating lies in the balance between rigid fixation and dynamic load sharing. Our engineers are currently exploring 3D-printed porous titanium structures that mimic the elastic modulus of human bone, reducing the "stress shielding" effect and improving long-term clinical outcomes for patients undergoing multi-level fusions.
Full regulatory dossiers provided for streamlined local registration in any jurisdiction.
Strategic partnerships with global carriers ensuring duty-efficient delivery to 80+ countries.
Custom design capabilities to meet specific anatomical requirements for different ethnic populations.
Dedicated clinical support team to assist with instrument handling and surgical planning.
Ensuring every implant exceeds international safety benchmarks.
 Fatigue Tester (ASTM F1717)
Fatigue Tester (ASTM F1717) Torsion Tester
Torsion Tester ONH Series Analyzer
ONH Series AnalyzerOur highly equipped R&D lab conducts relentless testing. For instance, our Fatigue Tester simulates years of human movement to ensure our plates never fail under physiological loads. The Torsion Tester guarantees that locking screws maintain their grip without stripping or loosening. This meticulous approach is why we are considered a top Trauma & Orthopedic Implants company worldwide.
We primarily use Grade 5 Titanium Alloy (Ti-6Al-4V ELI), which offers the best combination of biocompatibility, high strength-to-weight ratio, and excellent fatigue resistance for spinal applications.
Every plate and screw undergoes 100% inspection using high-resolution CNC optical measuring systems to ensure tolerances are kept within microns, preventing the common issue of "screw back-out."
Yes, we provide a complete technical file including biocompatibility reports, clinical evaluation reports, and manufacturing validation data to assist our partners in local regulatory filings.
Depending on order volume, our Industry 4.0 production lines allow us to maintain a standard lead time of 15-30 days, significantly faster than the industry average.
Explore our full range of trauma and spinal fixation systems.

Sports Medicine Interference Screws for Acl Reconstruction Peek and Cannulated Screws

Surgical Suture Lasso Medical Arthroscopy Instruments Suture Hooks for Labral Repair

Canwell Orthopedic Trauma Implant Ilizarov Ring Fixator External Fixator Ilizarov External Fixation


Plastic Peek Hexagon Allen Socket Head PVDF Screws Bolts Nuts Auto Fastener
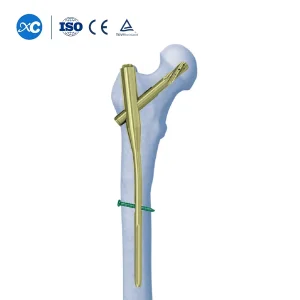
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
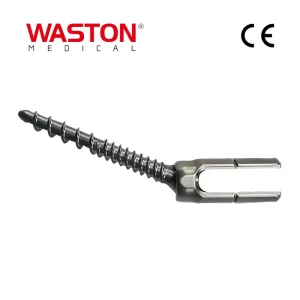

Orthopedic Instrument Ring Frame Ring Fixator Lower Limb Lengthening External Fixator

