 Orthopedic Surgery Trauma Manufacturer Finger External Fixator for Mini Fixator
Orthopedic Surgery Trauma Manufacturer Finger External Fixator for Mini Fixator
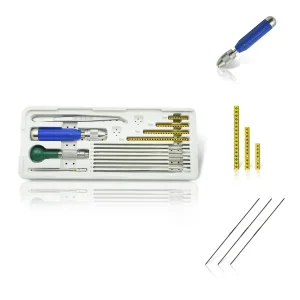 Veterinary Mini External Fixator for Birds, Reptiles & Small Mammals
Veterinary Mini External Fixator for Birds, Reptiles & Small Mammals
 Factory Direct Supply High Quality Small Fragment External Fixator
Factory Direct Supply High Quality Small Fragment External Fixator
 5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
 Ilizarov Talipes Equinovalgus Bone Lengthening External Fixator
Ilizarov Talipes Equinovalgus Bone Lengthening External Fixator
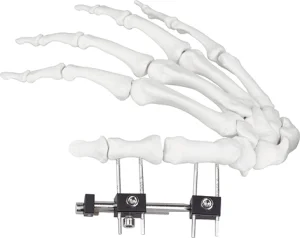 External Fixator Hand Bone Pin Stent Radius Mini Fixator
External Fixator Hand Bone Pin Stent Radius Mini Fixator
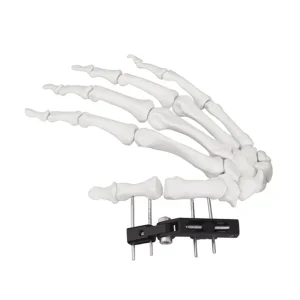 External Fixator Hand Bone Pin Stent Radius Mini Fixator Track Type
External Fixator Hand Bone Pin Stent Radius Mini Fixator Track Type
 Orthopedic External Fixator Mini Fragment for Finger and Palm
Orthopedic External Fixator Mini Fragment for Finger and Palm
The global orthopedic market is witnessing a paradigm shift, particularly in the realm of Spinal Internal Fixation. As the world's population ages and the incidence of spinal disorders—from degenerative disc diseases to complex trauma—rises, the demand for high-precision, biocompatible, and reliable fixation systems has reached unprecedented levels. Modern spinal surgery is no longer just about stabilization; it is about restoring biomechanical function and improving the quality of human life.
Today, internal fixators serve as the cornerstone for treating spinal instability. Whether it is through pedicle screw systems, spinal rods, or interbody cages, the industrial focus has moved toward minimally invasive surgery (MIS). This evolution requires manufacturers to not only master traditional metallurgy but also embrace advanced CAD/CAM modeling and robotic-integrated designs. As a leading hub for medical device manufacturing, China has positioned itself at the forefront of this industrial revolution, blending cost-effective production with world-class R&D.
Transitioning from pure stainless steel to medical-grade Titanium alloys (Ti6Al4V) and PEEK materials for superior biocompatibility and reduced MRI interference.
Development of fixators designed specifically for robotic-arm assistance, ensuring sub-millimeter precision in screw placement and rod contouring.
The rise of patient-specific implants (PSI) utilizing 3D metal printing to create porous structures that promote faster osseointegration.
International buyers, ranging from large-scale government health departments to private surgical clinics, look for a specific set of criteria when sourcing spinal internal fixators. The primary demand is Regulatory Compliance. Certificates such as CE marking, ISO 13485, and FDA registration are non-negotiable prerequisites for entry into the European and American markets.
Furthermore, supply chain resilience has become a critical factor. Hospitals require "Just-In-Time" delivery to manage inventory costs effectively. This is where Chinese factories excel, offering high-volume production capacities coupled with streamlined logistics. Procurement managers are also increasingly focused on the "System Completeness"—preferring vendors who provide not just the screws and rods, but also the specialized surgical instrumentation required for implantation.
We are most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government. We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing, and we operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe and USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price !! We believe human life is invaluable !! We understand your requirements, we recognize your needs! Come partner with us...


Our highly equipped R&D lab and stringent quality control standards ensure quality as per international standards. QC starts from the raw material stage.
Leveraging China's industrial ecosystem allows us to provide premium orthopedic solutions at a fraction of the cost of Western competitors without sacrificing quality.
With a presence in over 80 countries, we understand localized regulatory hurdles and provide full documentation support for seamless customs clearance.
Our products are engineered for diverse clinical environments. In Emergency Trauma Centers, our spinal fixation systems provide rapid stability for vertebral fractures. In Specialized Geriatric Clinics, we focus on low-profile designs that cater to osteoporotic bone structures, ensuring secure purchase where traditional screws might fail.
Moreover, we have expanded into Veterinary Orthopedics. As shown in our product range, we offer mini-fixators for avian and small mammal spinal trauma, a rapidly growing niche in global veterinary medicine. This versatility demonstrates our ability to adapt industrial manufacturing to specialized medical needs.
Quality Control and R&D: We have a highly equipped R&D lab and stringent quality control standards to ensure quality of our products as per international standards. Quality Control at Narang Medical starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into manufacture of the finished product.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. We are thus considered as a top Trauma & Orthopedic Implants company.
 Competitive Price Bone Lengthening External Fixator Instrument
Competitive Price Bone Lengthening External Fixator Instrument
 Orthopedic Surgical Mini Combined Type Fragment External Fixator
Orthopedic Surgical Mini Combined Type Fragment External Fixator
 Orthopedic Stainless Steel Femur Fixator
Orthopedic Stainless Steel Femur Fixator
 High-Quality External Fixator for Bone Fixation (Durable)
High-Quality External Fixator for Bone Fixation (Durable)
 Titanium Alloy Femur Fixator for Bone Alignment
Titanium Alloy Femur Fixator for Bone Alignment
 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System
 Ilizarov Ring External Fixator Ankle Joint External Fixator
Ilizarov Ring External Fixator Ankle Joint External Fixator
 Ilizarov Ring External Fixator Knee Joint External Fixator
Ilizarov Ring External Fixator Knee Joint External Fixator