High-Precision Titanium Implants & Surgical Instruments

Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set


Titanium Alloy Anatomical Hook Guide Pin for Anterior Cervical Plate Fixation Surgery

External Fixation Orthopedic Fixator Pelvic Fracture Surgical Instruments Manufacture

Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
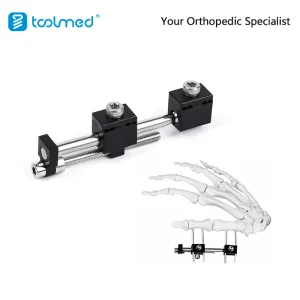
Small Bone External Fixator, Mini External Fixation System for Orthopedic Trauma

Analyzing market dynamics, technological breakthroughs, and the evolution of spinal stabilization.
The global market for Occipitocervical Fixation Systems is witnessing unprecedented growth, driven by a combination of aging demographics, an increase in spinal trauma cases, and the rapid advancement of surgical techniques. As a critical component of orthopedic surgery, these systems are designed to provide stabilization and fusion of the craniocervical junction, a complex anatomical region that demands extreme precision and biocompatibility.
Valued at billions globally, the demand for posterior cervical fixation is rising in North America, Europe, and Asia-Pacific, fueled by specialized neuro-trauma centers.
Shift from traditional stainless steel to Medical-Grade Titanium Alloys (Ti6Al4V) and PEEK materials to ensure MRI compatibility and superior bone ingrowth.
Integration with AI-driven surgical planning and 3D-printed custom patient-specific implants is the next frontier for "Smart Spinal Surgery."

We provide full-spectrum customization, from initial CAD design and rapid prototyping to mass production and sterile packaging.
Leveraging China's robust industrial supply chain, we offer premium quality implants at 30-50% lower costs than European counterparts.
Compliance with ISO 9001:2015 and EN ISO 13485:2016 ensures every screw and plate meets the rigorous demands of human clinical use.
Modern surgical trends are moving toward Minimally Invasive Surgery (MIS). Our research and development team is focused on developing cannulated poly-axial pedicle screws and low-profile occipital plates that reduce tissue trauma and speed up patient recovery. In global procurement, hospitals and large-scale medical distributors are increasingly looking for Custom OEM Occipitocervical Fixation System Factory partners who can provide not just the implants, but the specialized instrument sets required for complex procedures.
Localized application scenarios vary: In high-income regions, there is a push for Robotic-Assisted Spinal Surgery compatible implants. In developing markets, the focus remains on Durability, Ease of Use, and Affordability. As a top-tier supplier, we bridge this gap by offering a tiered product portfolio that caters to both high-tech neurosurgery centers and general trauma hospitals.
Committed to the sanctity of human life through superior medical manufacturing.
We are the most reputed manufacturers and exporters of Orthopedic Implants and Instruments, recognized as a Star Export House by the Government. Our presence spans over 80 countries worldwide, where we have built a reputation for superior quality and competitive pricing. We operate under a quality system that strictly complies with ISO 9001:2015, EN ISO 13485:2016, CE, and WHO: GMP Compliance. Many reputed companies in Europe and the USA regularly source their critical spinal components from our facility.
Unlike many competitors, we never compromise on quality for a lower price. We believe human life is invaluable. We understand your requirements and recognize the critical nature of these surgical tools. Quality Control at our factory starts at the raw material stage, allowing us to control the integrity of every single component that goes into the finished product.

Our R&D team consists of highly qualified engineers who persistently conduct research and seek feedback from the global community of orthopedic surgeons. This feedback loop allows us to create new designs that meet the future needs of Trauma and Orthopedic surgery. We aren't just a supplier; we are an innovation partner dedicated to advancing spinal health.
Precision Instruments for Minimally Invasive & Complex Surgeries