 Orthopedic Implants Cannulated Screw for Surgery Use
Orthopedic Implants Cannulated Screw for Surgery Use
 Orthopedic Medical Equipment Headless Cannulated Screw Instrument Set Surgical Instrument
Orthopedic Medical Equipment Headless Cannulated Screw Instrument Set Surgical Instrument
 Medical OEM&ODM Titanium Cannulated Headless Compression Screw
Medical OEM&ODM Titanium Cannulated Headless Compression Screw
 Headless Compression Hollow Screw Cannulated Bone Screws
Headless Compression Hollow Screw Cannulated Bone Screws
 Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm
Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm
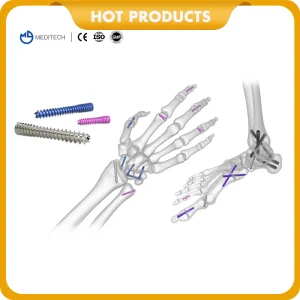 Orthopedic Implants Titanium Medical Bone Screws
Orthopedic Implants Titanium Medical Bone Screws
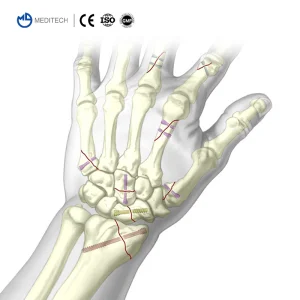 Titanium Medical Bone Screws Herbert Cannulated Compression Screw
Titanium Medical Bone Screws Herbert Cannulated Compression Screw
 Minimally Invasive Spinal Surgical Instrument Set Bone Surgery
Minimally Invasive Spinal Surgical Instrument Set Bone Surgery
🌐 The global orthopedic market is witnessing a paradigm shift toward Minimally Invasive Surgery (MIS). Cannulated screw instruments have become the cornerstone of modern trauma and reconstructive surgery. Designed with a central hollow core, these instruments allow surgeons to insert screws over a guide wire with pinpoint accuracy, significantly reducing tissue trauma and operative time. As an industry-leading Cannulated Screw Instrument Factory, we recognize that the demand is no longer just for hardware, but for comprehensive surgical solutions that ensure better patient outcomes.
The global orthopedic implant market is projected to reach billions by 2030, driven by an aging population and rising sports injuries. Cannulated screws dominate the fracture fixation segment due to their versatility in small and large bone procedures.
Modern surgery now integrates digital imaging and robotics. Our cannulated instruments are engineered to be compatible with advanced navigation systems, ensuring compatibility with the latest hospital infrastructures worldwide.
The shift from stainless steel to Grade 5 Titanium (Ti6Al4V) and bio-absorbable materials is a key trend. We prioritize Titanium for its superior biocompatibility and fatigue resistance.
🏭 As a premier Chinese manufacturer and exporter, we offer a unique blend of high-tech production capabilities and cost-efficiency that is difficult to match elsewhere. Our facility is equipped with state-of-the-art CNC machining centers, automated polishing lines, and clean-room packaging facilities. We don't just manufacture; we innovate.
Countries Exported
Certified Quality
Strict QC Inspection
Global Support
🏥 Cannulated screw instruments are essential in various surgical settings. From trauma centers in New York to specialized orthopedic clinics in Berlin, our products are used daily. Global procurement officers look for reliability, sterile integrity, and instrument ergonomics.
Ideal for femoral neck fractures, pelvic fractures, and epiphysiolysis. The cannulated design allows for minimally invasive entry, reducing post-operative pain.
In foot and ankle surgery (like scaphoid fractures or ankle fusion), precision is everything. Our headless compression screws provide the necessary torque without damaging soft tissue.
Quick-response instrument sets that are easy to sterilize and organize are in high demand for emergency trauma units worldwide.


We are most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government. We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing. Many reputed companies in Europe and USA regularly buy from us because we refuse to compromise.
"Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price !! We believe human life is invaluable !!"
We have a highly equipped R&D lab and stringent quality control standards to ensure quality of our products as per international standards. Quality Control at our facility starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into manufacture of the finished product.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. We are thus considered as a top Trauma & Orthopedic Implants company globally.
 Orthopedic Surgical Instrument Medical Femoral Interlocking Nail
Orthopedic Surgical Instrument Medical Femoral Interlocking Nail
 Cmf Maxillofacial Craniomaxillofacial Orthopedic Instrument
Cmf Maxillofacial Craniomaxillofacial Orthopedic Instrument
 Medical Orthopedic C Clamp Pelvic Instrument Set
Medical Orthopedic C Clamp Pelvic Instrument Set
 Surgical Trauma Minimally Invasive Spinal Mis Instrument
Surgical Trauma Minimally Invasive Spinal Mis Instrument
 Posterior Lumbar Interbody Fusion Cage System
Posterior Lumbar Interbody Fusion Cage System
 Orthopedic Anterior Cervical Instrument Set Medical
Orthopedic Anterior Cervical Instrument Set Medical
 Proximal Femoral Nail Instrument Sets Orthopedic Surgery
Proximal Femoral Nail Instrument Sets Orthopedic Surgery
 Xc Medico Orthopedic 6.0mm Spinal Pedicle Screw System
Xc Medico Orthopedic 6.0mm Spinal Pedicle Screw System