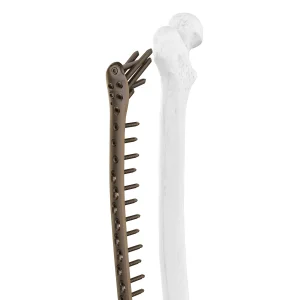
 Canwell Proximal Femoral Locking Plate Femur Plate Large Fragment Femoral Plate
Canwell Proximal Femoral Locking Plate Femur Plate Large Fragment Femoral Plate
 Titanium Plate Implant Price Surgical Medical Supply Orthopedic Implants Distal Femur Medial Condyle Locking Plate for Femur (L/R)
Titanium Plate Implant Price Surgical Medical Supply Orthopedic Implants Distal Femur Medial Condyle Locking Plate for Femur (L/R)
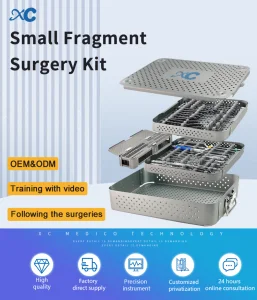 Orthopedic Surgical Trauma Implant Distal Radius and Proximal Humerus Locking Plate Instrument Small Fragment Set
Orthopedic Surgical Trauma Implant Distal Radius and Proximal Humerus Locking Plate Instrument Small Fragment Set
 Hot Selling Multi-Axial Distal Femur Lateral Locking Plate with CE
Hot Selling Multi-Axial Distal Femur Lateral Locking Plate with CE
 Variable Angle Orthopedic LCP Distal Femoral Locking Plate
Variable Angle Orthopedic LCP Distal Femoral Locking Plate
 Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation Bone Fracture Implants Medical Titanium Orthopedic Surgical Plate
Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation Bone Fracture Implants Medical Titanium Orthopedic Surgical Plate
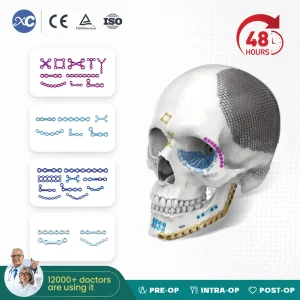 Cmf System Titanium Material Orthopedic Surgery Maxillofacial Locking Plates and Screws Implant
Cmf System Titanium Material Orthopedic Surgery Maxillofacial Locking Plates and Screws Implant
 Orthopedic Titanium Implants Distal Radius Locking Plate Instruments Set
Orthopedic Titanium Implants Distal Radius Locking Plate Instruments Set
The global orthopedic market has witnessed an unprecedented surge in demand for Distal Femur Locking Plates. As a critical component in internal fixation surgery, these plates are essential for treating complex supracondylar fractures of the femur. With the rise in the global geriatric population and the increasing incidence of high-energy trauma (such as traffic accidents), the industrial landscape for orthopedic implants has shifted towards high-precision, biocompatible solutions.
Leading healthcare systems in the United States, Europe, and Asia-Pacific are increasingly adopting Locking Compression Plate (LCP) technology. This technology combines the principles of compression plating and internal fixation, providing superior stability particularly in osteoporotic bone. As an OEM/ODM Distal Femur Locking Plate manufacturer, we sit at the intersection of medical engineering and industrial manufacturing, providing the hardware that allows surgeons to restore mobility and improve patient outcomes worldwide.
The transition from 316L Stainless Steel to Grade 5 Titanium Alloy (Ti6Al4V) is now standard, offering better fatigue strength and MRI compatibility.
Advanced Variable Angle (VA) locking technology allows surgeons to direct screws into specific anatomical zones, accommodating unique fracture patterns.
Research is pivoting toward integrating sensors into locking plates to monitor bone healing and load distribution in real-time.
When international hospitals and medical distributors source Distal Femur Locking Plates, they look for more than just a piece of metal. The procurement criteria are stringent, focusing on Regulatory Compliance (FDA, CE, ISO), Material Traceability, and Instrumentation Compatibility.
Global buyers require a reliable supply chain that can offer ODM (Original Design Manufacturing) services to customize plate geometry for local anatomical variations. For instance, populations in Southeast Asia may require smaller anatomical contours compared to European demographics. Our factory excels in providing these tailored solutions, ensuring that every plate fits perfectly, reducing surgical time and improving the surgical success rate.
Scenario 1: Geriatric Trauma Centers
In aging societies like Japan and Germany, "periprosthetic fractures" (fractures around a knee replacement) are common. Our distal femur locking plates are designed with specific screw hole patterns to bypass existing intramedullary components.
Scenario 2: High-Energy Trauma Units
In developing urban centers where construction and traffic accidents are frequent, surgeons require robust, multi-axial locking plates that can stabilize comminuted (shattered) fractures while allowing early mobilization.
Scenario 3: Minimal Invasive Surgery (MIS)
Modern surgeons prefer the MIPO (Minimally Invasive Percutaneous Osteosynthesis) technique. Our plates are designed with tapered ends and compatible insertion guides to facilitate small-incision surgery, reducing soft tissue damage and speeding up recovery.
We utilize 5-axis CNC machining centers from Switzerland and Japan to ensure micron-level precision for every locking hole and thread.
By leveraging China's integrated industrial supply chain, we offer premium medical devices at a fraction of the cost of Western competitors without sacrificing quality.
From initial CAD design to rapid prototyping and final sterile packaging, we provide a full-service turnkey solution for international brands.
We are most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government.
We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing, and we operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe and USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price !! We believe human life is invaluable !! We understand your requirements, we recognize your needs! Come partner with us...
We have a highly equipped R&D lab and stringent quality control standards to ensure quality of our products as per international standards. Quality Control at Narang Medical starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into manufacture of the finished product.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. We are thus considered as a top Trauma & Orthopedic Implants company.


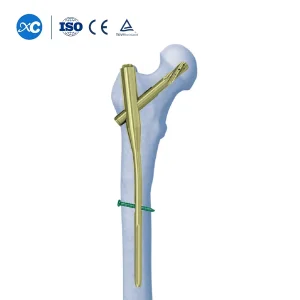 Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
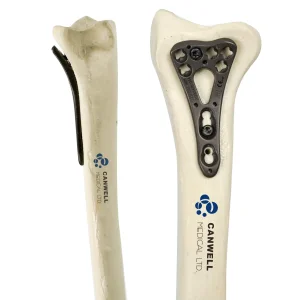 Canwell Variable Angle Radius Locking Plate Orthopaedic Implants Titanium Locking Plate
Canwell Variable Angle Radius Locking Plate Orthopaedic Implants Titanium Locking Plate
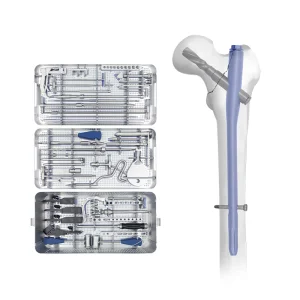 Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set
Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set
 Variable Angle Two-Column Volar Distal Radius Locking Compression Bone Plate, Va LCP System, Titanium Orthopedic Surgical Implant for Trauma Surgery, CE&ISO
Variable Angle Two-Column Volar Distal Radius Locking Compression Bone Plate, Va LCP System, Titanium Orthopedic Surgical Implant for Trauma Surgery, CE&ISO
 Locking Plate Multi Axial for Distal Radius Fracture Orthopedic Surgery Implants
Locking Plate Multi Axial for Distal Radius Fracture Orthopedic Surgery Implants
 Orthopedic Implants Locking Plate Multi Axial Distal Radius Cheap Price, Orthopedic, Medical Equipment
Orthopedic Implants Locking Plate Multi Axial Distal Radius Cheap Price, Orthopedic, Medical Equipment
 Locking Compression Plate (LCP) for Femur - LC-LCP Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
Locking Compression Plate (LCP) for Femur - LC-LCP Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
 Orthopedic Plates Titanium Medical Surgical Fracture Bone Trauma Large Fragment Proximal Femur Neck Plate with Hook Locking Plate with CE
Orthopedic Plates Titanium Medical Surgical Fracture Bone Trauma Large Fragment Proximal Femur Neck Plate with Hook Locking Plate with CE