 Minimally Invasive Spinal Mis Instrument Set Surgical Instrument Set for Bone Surgery Medical Device Spine System
Minimally Invasive Spinal Mis Instrument Set Surgical Instrument Set for Bone Surgery Medical Device Spine System Orthopedic Medical Equipment Headless Cannulated Screw Instrument Set Surgical Instrument
Orthopedic Medical Equipment Headless Cannulated Screw Instrument Set Surgical Instrument Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set
Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set Orthopedic Medical Equipment Surgical Trauma Minimally Invasive Spinal Mis Instrument Set
Orthopedic Medical Equipment Surgical Trauma Minimally Invasive Spinal Mis Instrument Set 5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws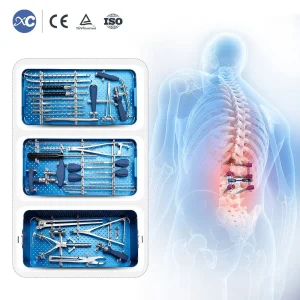 Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set Orthopedic Surgical Instrument Medical Femoral Interlocking Nail Instrument Set
Orthopedic Surgical Instrument Medical Femoral Interlocking Nail Instrument SetThe global spinal surgery market has transitioned into a new era of precision medicine. As a leading Spinal Surgery Instrument Manufacturer, we recognize the shifting dynamics from traditional open surgeries to complex, robot-assisted procedures. The market is currently driven by a rising geriatric population, the increasing prevalence of spinal disorders, and a massive demand for more effective, less invasive treatment options.
In industrial terms, the manufacturing of spinal instruments involves the integration of biocompatible materials like Titanium alloy (Ti6Al4V) and PEEK (Polyether ether ketone). Our role as an OEM/ODM supplier is to bridge the gap between clinical requirements and engineering possibilities. Hospitals across North America, Europe, and Asia-Pacific are increasingly seeking instruments that offer higher tactile feedback, reduced surgical time, and superior long-term patient outcomes.

The "Gold Standard" in spinal care has evolved toward Minimally Invasive Spinal Surgery (MISS). This trend focuses on minimizing muscle tissue disruption, reducing blood loss, and accelerating post-operative recovery. As a specialized factory, we have invested heavily in R&D to produce specialized tubular retractors, cannulated screw systems, and endoscopic instruments that allow surgeons to perform complex fusions through incisions smaller than 2cm.
Our instruments are designed to be compatible with advanced intraoperative imaging, such as O-arm and C-arm systems. This technological synergy ensures that pedicle screw placement is accurate to the sub-millimeter level, significantly lowering the risk of neurological complications.
Global procurement teams from leading medical device brands are increasingly looking toward China for high-value OEM partnerships. Why? Because we offer a unique combination of scale, speed, and sophistication. As a premier China-based factory, we provide:
By optimizing our supply chain and leveraging domestic high-grade titanium resources, we provide competitive pricing that allows distributors to maintain healthy margins while offering hospitals affordable solutions.
Have a custom design? Our engineers can take a CAD model to a functional prototype in record time, utilizing 5-axis CNC machining and AI-assisted design verification.
We understand that navigating the FDA, MDR (Medical Device Regulation), and local MOH registrations is complex. Our documentation is audit-ready, ensuring a smooth path to market entry for our partners.
Spinal instruments aren't just tools; they are extensions of the surgeon's hands. In Degenerative Disc Disease (DDD), our interbody fusion cages provide the necessary height restoration and stability. In Spinal Trauma, our posterior fixation systems offer immediate rigid stabilization, vital for neurological recovery.
Furthermore, our instrumentation is optimized for localized needs. For instance, in aging populations with Osteoporosis, we offer specialized fenestrated screws for cement augmentation, providing superior pull-out strength in compromised bone quality.

We are most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government.
We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing, and we operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe and USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price !! We believe human life is invaluable !! We understand your requirements, we recognize your needs! Come partner with us...
We have a highly equipped R&D lab and stringent quality control standards to ensure quality of our products as per international standards. Quality Control starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into manufacture of the finished product.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. We are thus considered as a top Trauma & Orthopedic Implants company.
The journey of spinal instrumentation began with basic rods and wires but has transformed into a sophisticated biomechanical field. Modern Spinal Surgery Instrument Manufacturers must account for the complex physics of the human spine—balancing rigidity with flexibility. The introduction of the pedicle screw revolutionized the industry by providing a three-column fix that was previously impossible. This advancement allowed for more aggressive corrections of spinal deformities and faster mobilization of patients. As a supplier, we ensure that our pedicle screws feature a double-lead thread design for faster insertion and a low-profile head to minimize soft tissue irritation.
In the world of OEM/ODM Spinal Instruments, material choice is paramount. Titanium remains the favorite for fixation due to its incredible strength-to-weight ratio and its ability to osseointegrate—where the bone actually grows into the metal. However, for interbody cages, PEEK has become the industry standard because its modulus of elasticity is similar to human cortical bone, reducing the risk of "stress shielding." Our factory utilizes medical-grade raw materials imported from certified suppliers in Germany and the USA, ensuring that every screw, plate, and rod meets the highest biocompatibility standards.
The recent global shifts have highlighted the importance of a resilient and transparent supply chain. Procurement officers are no longer just looking at price; they are looking at lead times and reliability. Our facility in China acts as a hub that mitigates these risks. By maintaining a vast inventory of standard components and having an agile manufacturing floor, we can pivot between large-scale OEM orders and specialized, small-batch ODM requests. This flexibility is what distinguishes a mere vendor from a true strategic manufacturing partner.
We are entering the era of "Surgery 4.0." This involves the use of artificial intelligence to plan surgeries before the patient even enters the OR. As an innovative manufacturer, we are developing "Smart Instruments" that can communicate with navigation systems. Imagine a torque wrench that automatically logs the exact Newton-meters applied to a screw and uploads it to the patient’s digital record. While this sounds like science fiction, it is the direction our R&D team is heading, ensuring that our partners are always at the forefront of medical technology.
For any Spinal Surgery Instrument Supplier, compliance is the ultimate barrier to entry. The transition from MDD to MDR in Europe has raised the bar for clinical evidence and traceability. Our quality management system is designed to provide full "cradle-to-grave" traceability for every instrument. We provide our clients with comprehensive technical files, including biocompatibility reports, sterilization validation, and clinical evaluation reports (CERs). This commitment to documentation is why our products are trusted by surgeons in over 80 countries, including the highly regulated markets of Western Europe.
The best instrument in the world is useless if not used correctly. As part of our service, we provide comprehensive surgical technique guides and video tutorials for our instrument sets. We also offer "White Label" educational materials for our OEM partners, allowing them to provide value-added service to their surgical clients. Our commitment to education ensures that the clinical outcomes reflect the quality of the instruments we manufacture.
Choosing a Spinal Surgery Instrument Manufacturer is a decision that impacts human lives. It requires a partner that values precision as much as the surgeon does. At our core, we are a blend of traditional craftsmanship and futuristic technology. Whether you are looking to launch a new line of MIS screws or seeking a reliable supplier for trauma sets, we have the expertise, the capacity, and the heart to bring your vision to life. Let us build the future of spinal care together.
 Proximal Femoral Nail Instrument Sets for Orthopedic Surgery Medical Implement
Proximal Femoral Nail Instrument Sets for Orthopedic Surgery Medical Implement Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set Cmf Maxillofacial Craniomaxillofacial Orthopedic Medical Instrument Set Surgical
Cmf Maxillofacial Craniomaxillofacial Orthopedic Medical Instrument Set Surgical Medical Orthopedic C Clamp Pelvic Instrument Set Surgical Instrument
Medical Orthopedic C Clamp Pelvic Instrument Set Surgical Instrument Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set for Knee Reconstruction
Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set for Knee Reconstruction Orthopedic Medical Equipment Posterior Lumbar Interbody Fusion Cage System (Straight type) Surgical Instrument Set
Orthopedic Medical Equipment Posterior Lumbar Interbody Fusion Cage System (Straight type) Surgical Instrument Set Acl Pcl Surgery Instrument Orthopedic Metal Arthroscopy Suture Anchors
Acl Pcl Surgery Instrument Orthopedic Metal Arthroscopy Suture Anchors Orthopedic Anterior Cervical Instrument Set Medical Instruments
Orthopedic Anterior Cervical Instrument Set Medical Instruments