The global market for Tibial Intramedullary (IM) Nails is witnessing unprecedented growth, driven by a rising incidence of road traffic accidents, sports-related injuries, and an aging population prone to osteoporotic fractures. As the "gold standard" for treating long bone diaphyseal fractures, the demand for high-precision, biocompatible, and easy-to-insert nailing systems has never been higher.
Technological advancements in the industrial sector have allowed OEM/ODM Tibial Intramedullary Nail Factories to shift from standard manufacturing to high-tech customized solutions. Today, surgeons and procurement specialists look beyond just the implant; they seek integrated systems that include specialized instrument sets, locking mechanisms, and innovative approaches like the suprapatellar technique.

The shift towards suprapatellar approach allows for easier fracture reduction and reduced pain, requiring specialized instrumentation that we provide as an OEM leader.
Utilizing AI to optimize the anatomical curvature of the tibial nail ensures a better fit across diverse ethnic populations, reducing "bursting" risks at the distal end.
Advanced anodization and sandblasting techniques enhance osseointegration and reduce the risk of post-operative infections in trauma patients.

Successful orthopedic surgery relies as much on the instruments as it does on the implant. Our Tibial Intramedullary Nail Instrument Set is designed for ergonomic efficiency and extreme durability. Featuring high-grade stainless steel components, our sets allow surgeons to perform complex interlocking procedures with minimal surgical time.
As a leading ODM partner, we can customize these sets to include color-coded guides, specialized reamers, and carbon-fiber aiming arms to facilitate better fluoroscopic visibility during surgery.
Professional buyers from medical distribution companies, government hospitals, and private trauma centers require suppliers who understand the nuances of localized applications and regulatory hurdles.
For distributors in Europe and the USA, we provide full CE and WHO: GMP compliance, ensuring that every batch of nails meets the most stringent safety standards.
In developing regions, we offer cost-effective "Expert" series nails that provide high clinical outcomes without the premium price tag of Big-Tech brands.
Our titanium nails are engineered for high-impact trauma centers where speed and reliability are critical. Our locking mechanisms ensure immediate stability.
The Chinese medical device manufacturing sector has evolved into a global powerhouse of innovation and efficiency. Choosing us as your Tibial Intramedullary Nail Supplier offers distinct advantages:

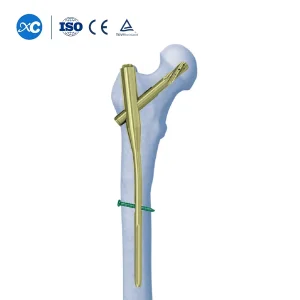
While we specialize in Tibial nails, our Proximal Femoral Nail Antirotation (PFNA) systems represent the pinnacle of our trauma engineering. Designed for trochanteric fractures, these nails utilize a helical blade that achieves superior bone compression and rotational stability.
Check out our Titanium Femur Intramedullary Nail solutions for comprehensive trauma management portfolios. Our OEM capability allows us to offer varying diameters and lengths to suit local surgical preferences.
We are most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government.
We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing, and we operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe and USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price !! We believe human life is invaluable !! We understand your requirements, we recognize your needs! Come partner with us...
We have a highly equipped R&D lab and stringent quality control standards to ensure quality of our products as per international standards. Quality Control at Narang Medical starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into manufacture of the finished product.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. We are thus considered as a top Trauma & Orthopedic Implants company.