 Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
Titanium Orthopedic Implants Fixation Surgical Cannulated Screw
 High Quality Orthopedic Titanium Metal Cancellous Cannulated Screws Stainless Steel Full-Thread Self-Tapping Bone Screw
High Quality Orthopedic Titanium Metal Cancellous Cannulated Screws Stainless Steel Full-Thread Self-Tapping Bone Screw
 Orthopedic Cannulated Screw for Bone Fixation Trauma Implant
Orthopedic Cannulated Screw for Bone Fixation Trauma Implant
 Titanium Scew, Headless Screw, Compression Screws, Surgical Instrument, Cannulated Screw
Titanium Scew, Headless Screw, Compression Screws, Surgical Instrument, Cannulated Screw
Switzerland stands as a global titan in the Medical Technology (MedTech) sector. As a primary Cannulated Screws Manufacturer & Supplier, the Swiss market demands nothing less than perfection. Cannulated screws, characterized by their hollow central shaft, are vital components in modern orthopedic surgery, allowing for precise placement over a guide wire. In the industrial heartlands of Zurich, Basel, and Geneva, the integration of advanced metallurgy and robotic precision has set a new benchmark for these life-saving implants.
The Swiss orthopedic landscape is driven by a unique combination of high-tech innovation and a robust healthcare infrastructure. Whether it's treating complex fractures in the elite ski resorts of the Swiss Alps or providing geriatric care in urban centers, our cannulated screws are engineered to meet the most rigorous clinical requirements.

Utilizing Grade 5 Titanium and premium Stainless Steel, ensuring maximum biocompatibility and osseointegration for Swiss patients.
Hollow-core technology with tolerances measured in microns, facilitating minimally invasive surgical procedures.
Designed with advanced cutting flutes that reduce insertion torque and minimize bone thermal necrosis during surgery.
The Swiss MedTech industry contributes significantly to the national GDP, with a heavy emphasis on export quality. Recent trends in the Swiss Cannulated Screw market highlight a shift toward "Smart Implants." Engineers are now exploring the integration of sensor technology within the cannulated core to monitor healing progress and local strain.
Switzerland's unique geography plays a role in the high demand for trauma implants. High-velocity sports injuries in mountain regions necessitate immediate and stable fixation of femoral neck fractures and ankle syndesmosis injuries. Our Cannulated Compression Screws are the preferred choice for Swiss trauma surgeons due to their ability to provide interfragmentary compression with minimal soft tissue disruption.
In the industrial sectors of Schaffhausen and Solothurn, the focus is on the "Industry 4.0" approach. This includes 3D-printed titanium screws and customized implant geometries based on CT scans, a service increasingly sought after by top-tier Swiss clinics like Hirslanden and University Hospital Zurich.

We are the most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by the Government. Our presence in the Swiss market is backed by a commitment to excellence that mirrors the Swiss "Watchmaking" precision.
We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing. We operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe (including major Swiss distributors) and the USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price! We believe human life is invaluable! We understand your requirements, we recognize your needs! Come partner with us for the most reliable cannulated screw solutions in Switzerland.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. Quality Control starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into the manufacture of the finished product. We are thus considered as a top Trauma & Orthopedic Implants company serving the Swiss medical community.
When selecting a Cannulated Screws Supplier in Switzerland, hospitals and distributors look for specific technical advantages. Our screws feature:
The industrial status of bone screw manufacturing has evolved. In Switzerland, there is an increasing demand for "Green MedTech." Our manufacturing processes emphasize sustainable energy use and minimal waste, aligning with the environmental goals of the Swiss confederation.
 Hb7.3 Titanium or Ss Cannulated Compression Screws
Hb7.3 Titanium or Ss Cannulated Compression Screws
 Interference Screws for Acl Reconstruction Peek and Cannulated Screws Sports Medicine
Interference Screws for Acl Reconstruction Peek and Cannulated Screws Sports Medicine
 Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants Bone Implant
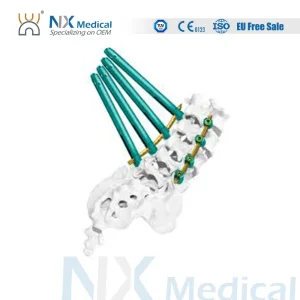 Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw 5.5mm System
 Jin Medical Orthopedic Small Cannulated Screw with CE
Jin Medical Orthopedic Small Cannulated Screw with CE
 Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
Orthopedic Implants Titanium Medical Bone Screws Cannulated Headless Compression Screw
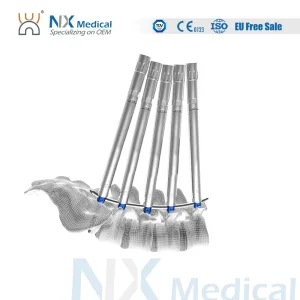 Nx Medical Spine Implant Cannulated Bone Cement Pedicle Screw for Spine Deformity
Nx Medical Spine Implant Cannulated Bone Cement Pedicle Screw for Spine Deformity
 Medical Cement Spine Cannulated Pedicle Screw
Medical Cement Spine Cannulated Pedicle Screw
Choosing the right Cannulated Screws Manufacturer involves evaluating more than just the product; it's about the partnership. In the competitive Swiss market, where precision is a cultural heritage, we provide implants that undergo multi-stage quality checks. Our presence as a leading Cannulated Screws Supplier in Switzerland is bolstered by our ability to offer competitive pricing without sacrificing the "Swiss-level" quality expected by surgeons in Bern, Lucerne, and beyond.
From Herbert screws used in scaphoid fractures to large 7.3mm screws for femoral neck fixation, our catalog is comprehensive. We support Swiss distributors with full documentation, including CE certifications and clinical evaluation reports, ensuring a smooth path through Swissmedic regulations.