











| Still deciding? Get samples of $ ! US$ 119/Piece Request Sample |


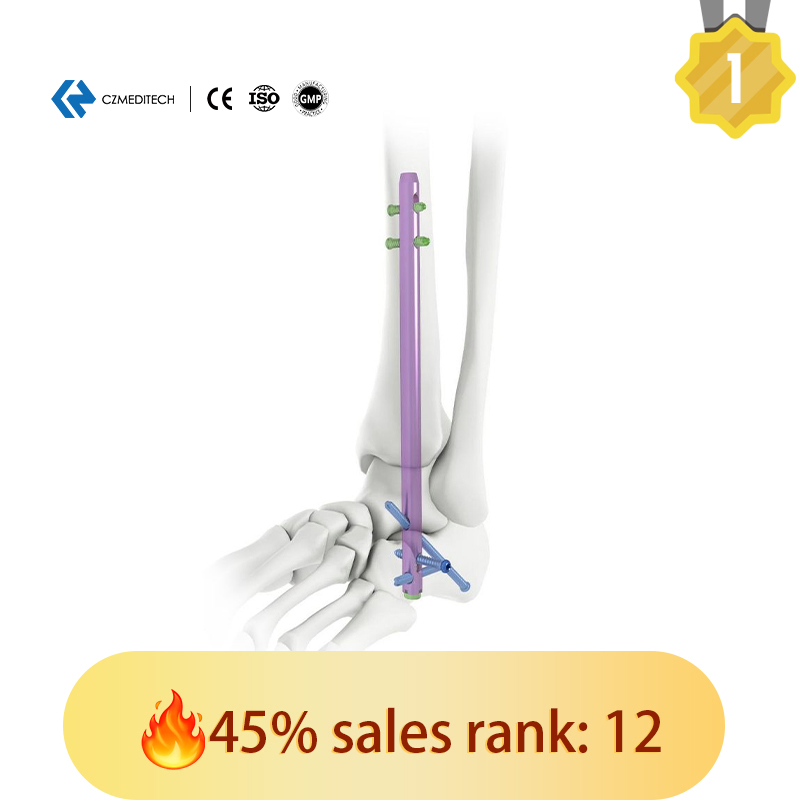
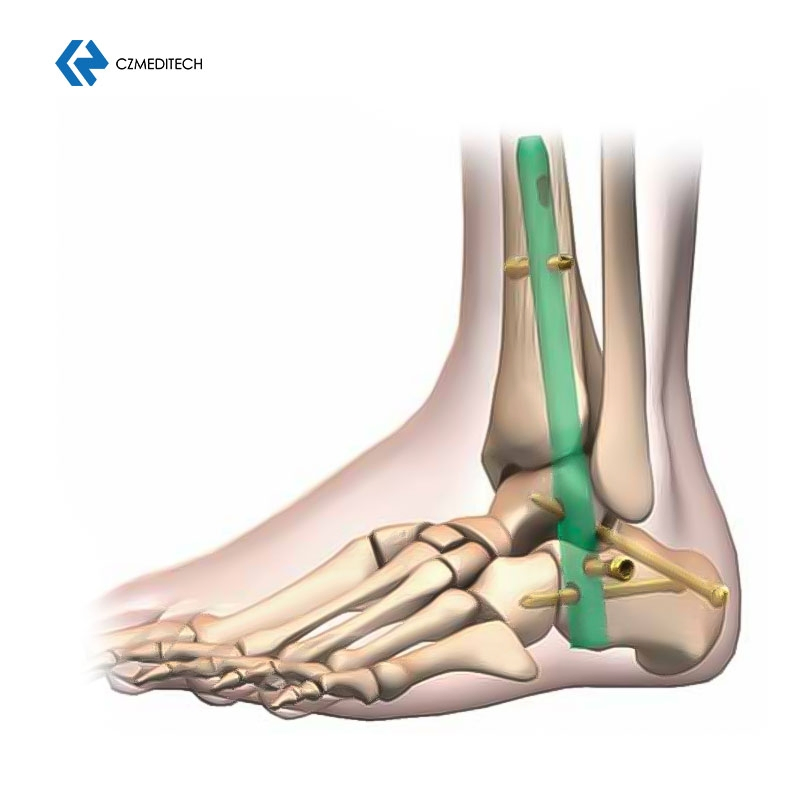
This titanium intramedullary nail is specifically designed for hindfoot and ankle fusion procedures, including tibiotalocalcaneal (TTC) arthrodesis. The interlocking design provides excellent rotational and axial stability, promoting successful fusion. Made from medical-grade titanium alloy (Ti6Al4V), it offers superior biocompatibility, strength, and MRI compatibility. Available in multiple sizes to accommodate patient anatomy.

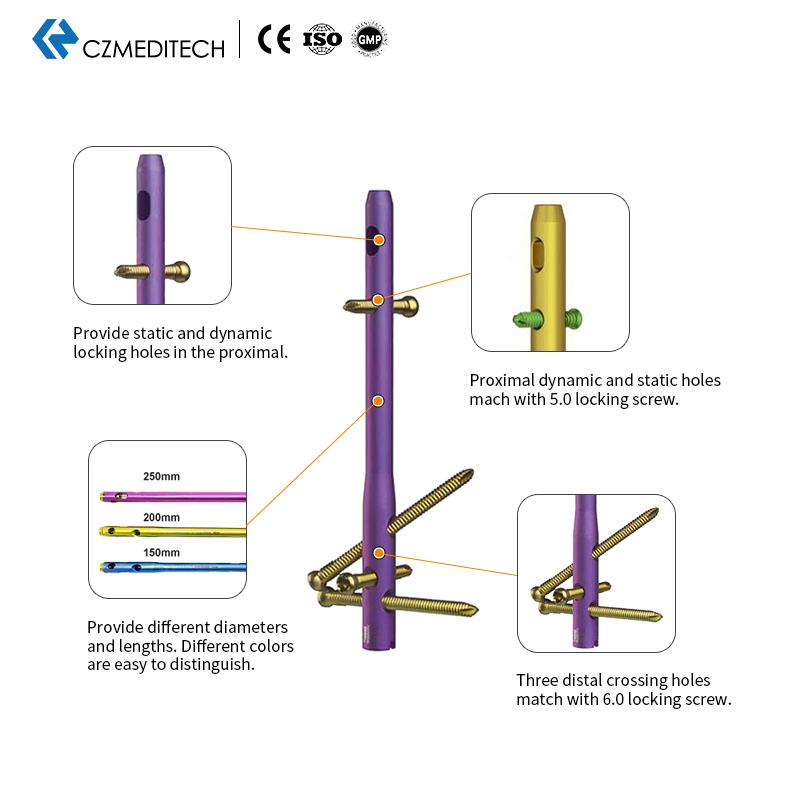
Available in different diameters and lengths. Color-coded for easy identification.

Provides static and dynamic locking holes in the proximal section.

Proximal dynamic and static holes match with 5.0 locking screw.

Three distal crossing holes match with 6.0 locking screw.
| Reference Item | Description / Dimensions |
|---|---|
| Hindfoot Fusion Ankle Nail | 10*150/200/225/250 mm (Left) |
| 10*150/200 mm (Right) | |
| 11*150/200/250 mm (Left) | |
| 11*150/200/225/250 mm (Right) | |
| 12*150/200/250 mm (Left) | |
| 12*150/200/250 mm (Right) | |
| 5.0mm Locking Screws | Length: 28mm to 60mm (2mm increments) |
| 6.0mm Locking Screws | Length: 26mm to 80mm (variable increments), up to 100mm |
As a leader in orthopedic implants and instruments manufacturing, we have been successfully supplying to over 2,500 clients in 70+ countries for over 15 years.

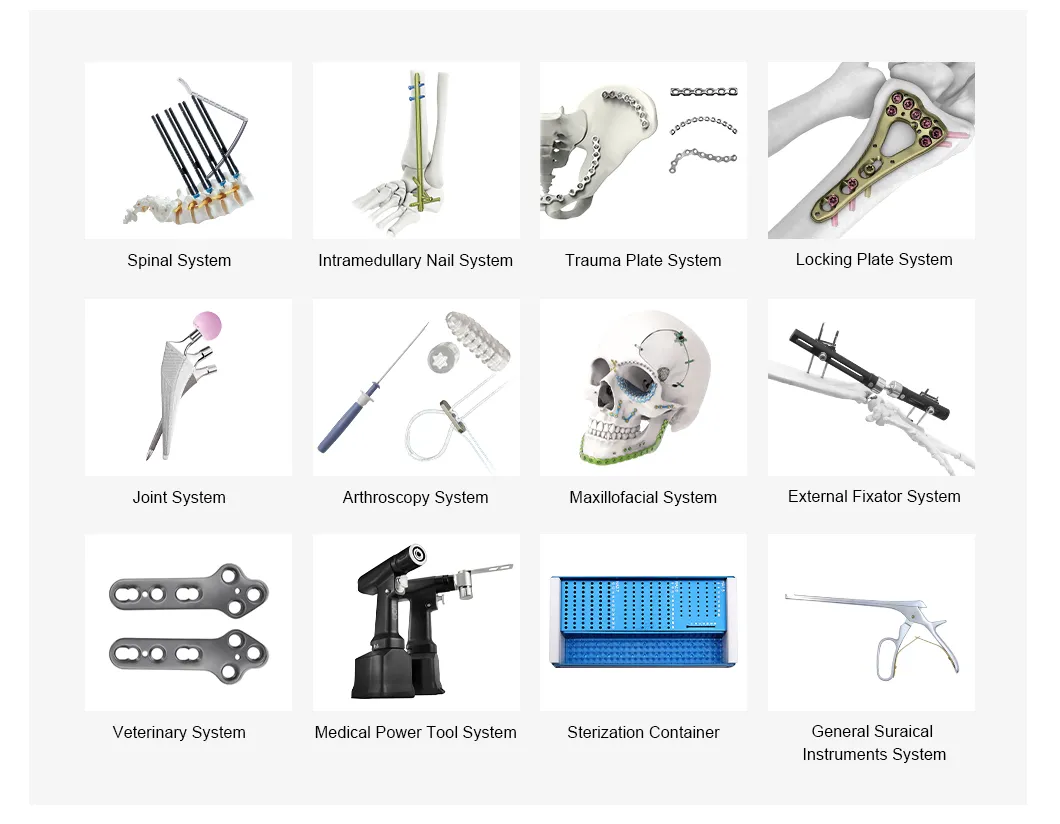
A comprehensive product portfolio spanning orthopedics, trauma care, veterinary medicine, and sterilization.

We provide a 1-year warranty. During the warranty period, we repair or replace products free of charge. Out-of-warranty repairs are charged at reasonable costs. All complaints receive responses within 2 days.
Certified Quality: We are CE certified and ISO 13485 quality management system certified.

ARAB HEALTH
UAE

MEDICA
Germany

HOSPITAL EXPO
Indonesia
 Voila Orthopedic Implants
Voila Orthopedic Implants