




1 / 5










| Still deciding? Get samples of $ ! US$ 50/Piece Request Sample |
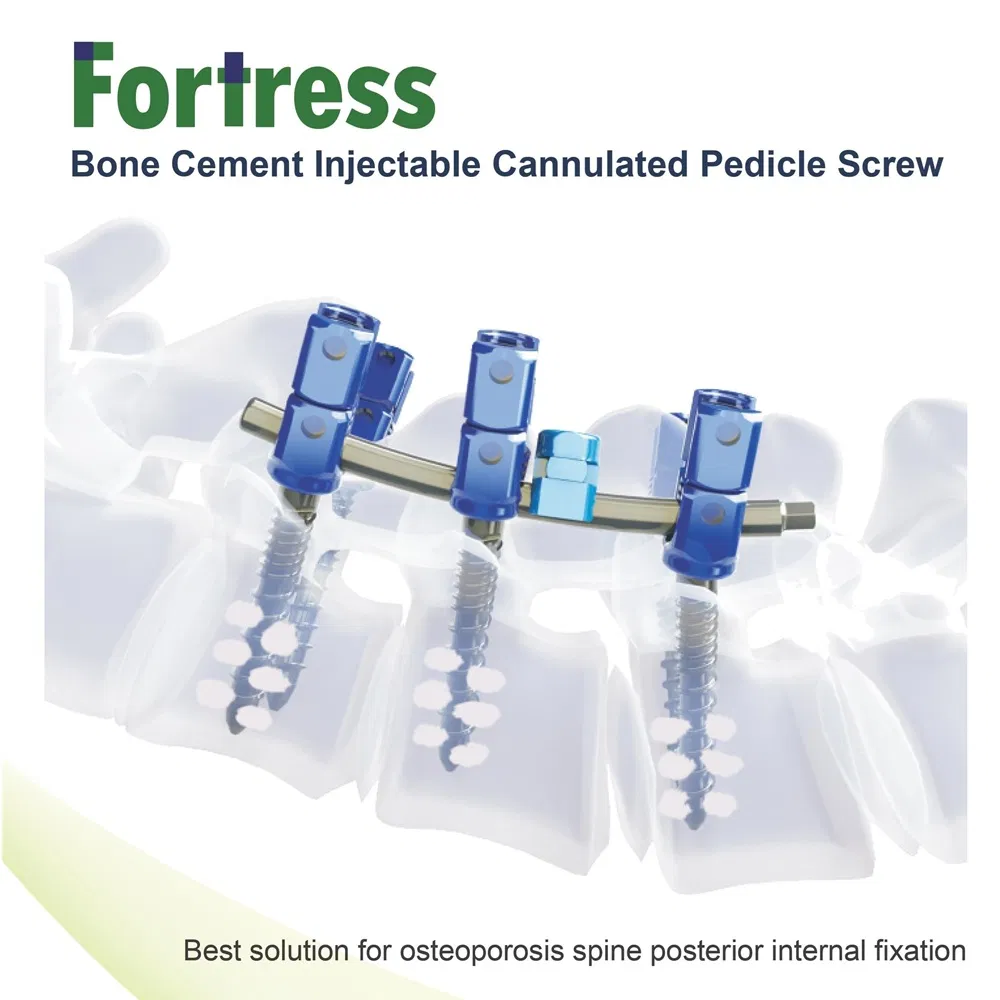
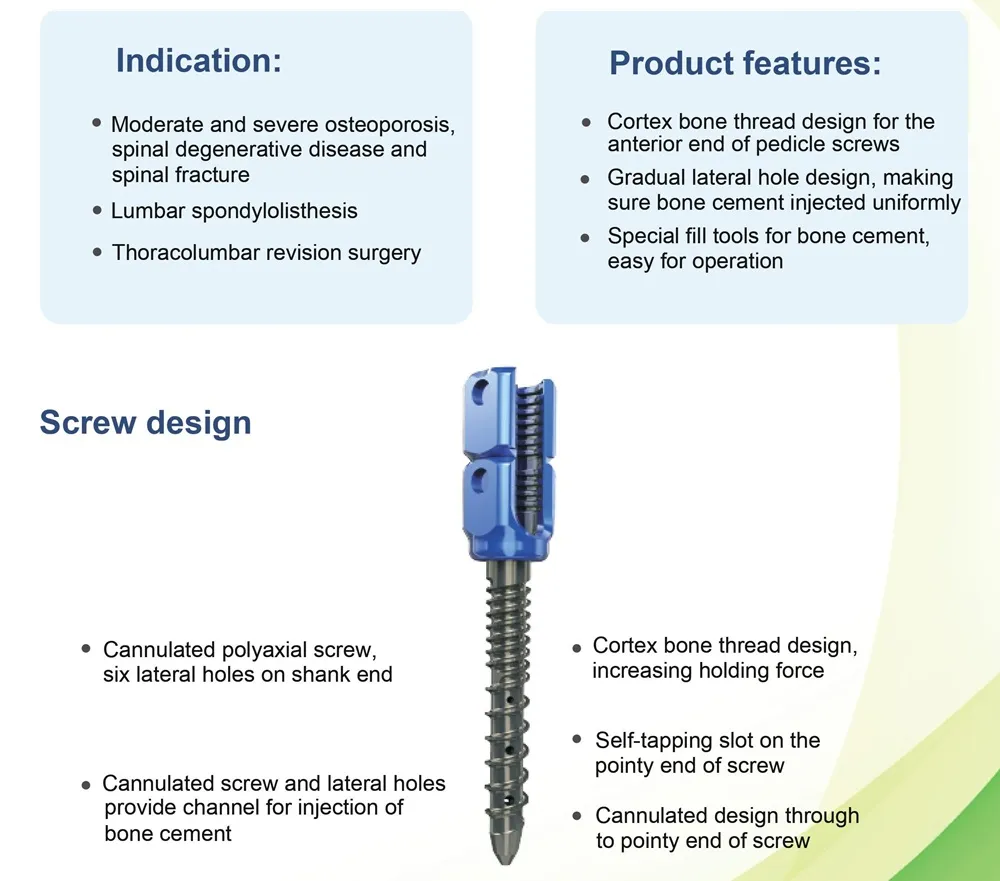
This advanced orthopedic solution is specifically designed for spinal stability in complex surgeries, featuring a specialized delivery system for bone cement.
| Parameter | Details |
|---|---|
| Model NO. | DF-II VIII |
| Application | Orthopedic Surgery |
| Material | Titanium Alloy |
| Product Name | Bone Cement Cannulated Pedicle Screw |
| Diameter Range | 4.0 / 4.5 / 5.0 / 5.5 / 6.0 / 6.5 / 7.0 mm |
| Length Range | 25-60 mm |
| Production Capacity | 10,000 PCS/Year |
| HS Code | 90211000 |
| Origin | China |
| Product Name | Specification (Diameter & Length) |
|---|---|
| Reduction Monoaxial Bone Cement Pedicle Screw | Diameter: 4.0/4.5/5.0/5.5/6.0/6.5 mm Length: 25-60 mm |
| Reduction Polyaxial Bone Cement Pedicle Screw | Diameter: 4.0/4.5/5.0/5.5/6.0/6.5/7.0 mm Length: 25-60 mm |


- Implants: Inner plastic bag + outer carton box.
- Instruments Set: Inner instruments box + outer carton box.
- Customized packing design available.
- International Air Freight
- Express: DHL, UPS, FedEx, etc.

One-stop orthopedic product solution provider since 2016.



 Voila Orthopedic Implants
Voila Orthopedic Implants