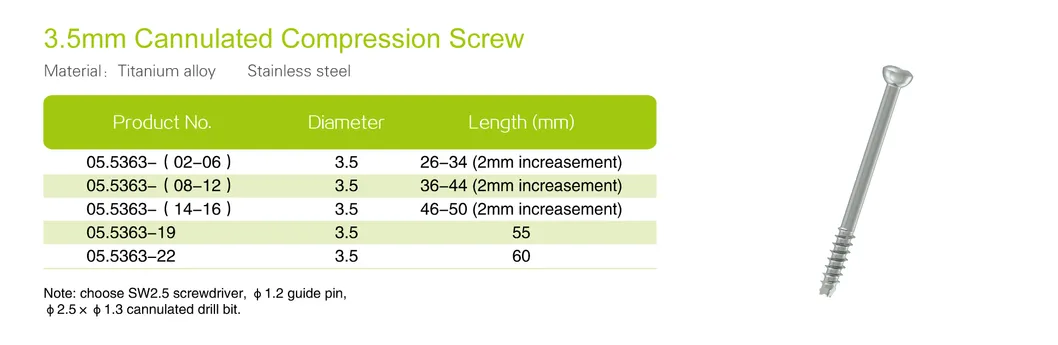
Cannulated screws are a common method of fixation used by orthopedic surgeons for multiple fracture patterns. These screws are "hollow" to allow them to be placed over a guidewire facilitating better alignment prior to drilling or screw insertion. The guidewire is then removed and the screw is left in place.
Available in headless and with-head types, with various sizes including 3.0, 4.0, 6.5, and 7.3mm.
✔
Superior Material: Titanium alloy material has better biocompatibility; compatible with CT and MRI.
✔
Tissue Protection: Lower nail head notch reduces interference with surrounding tissue.
✔
Versatile Design: Available in 1/3 thread, half thread, and full thread designs.
✔
Efficient Operation: Self-tapping and self-drilling design effectively shortens operation time.
✔
Precision Fixation: Torx head provides better control and prevents skidding.
✔
Stable Compression: Maintains compression function under periodic forces after insertion.
? Frequently Asked Questions
1. What material are these cannulated compression screws made of?
They are made of high-quality Titanium alloy, which offers excellent biocompatibility and is compatible with CT and MRI imaging.
2. What are the available thread options for the 7.3mm screw?
We provide three different thread designs: 1/3 thread, half thread, and full thread to suit various clinical needs.
3. How does the Torx head design benefit the surgical procedure?
The Torx head allows for better torque transmission and control, significantly reducing the risk of screwdriver skidding during insertion or removal.
4. Can these screws be used for emergency trauma cases?
Yes, the self-tapping and self-drilling design makes them easy to operate and helps to shorten the overall operation time in critical procedures.
5. What sizes are available for these orthopedic cannulated screws?
Besides the 7.3mm diameter, cannulated screws are typically available in 3.0, 4.0, and 6.5mm sizes to accommodate different fracture patterns.
6. Are the products supplied sterile?
The screws can be provided in either Sterile or Non-Sterile packaging depending on the specific requirement and facility protocols.























 Voila Orthopedic Implants
Voila Orthopedic Implants