



1 / 5










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |
Matching Screws:
Matching Instruments:

| Model Name | Specification Details |
|---|---|
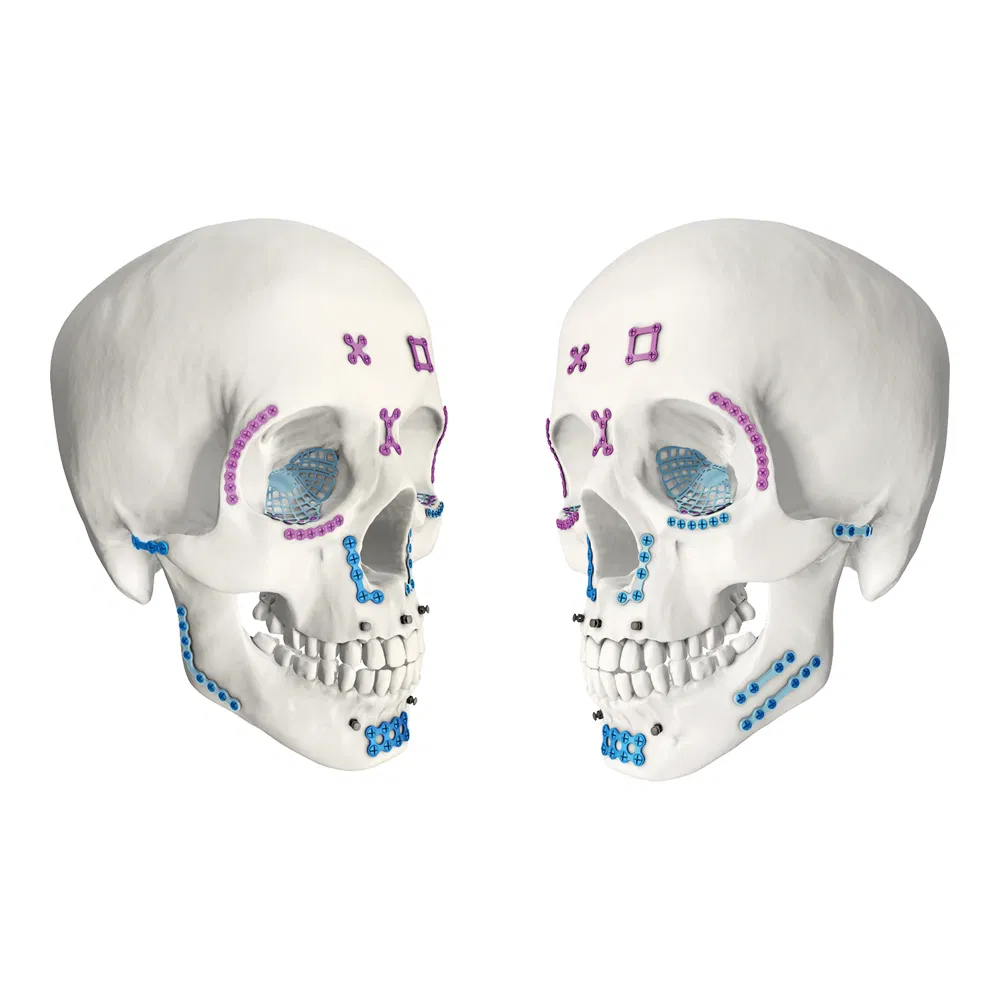
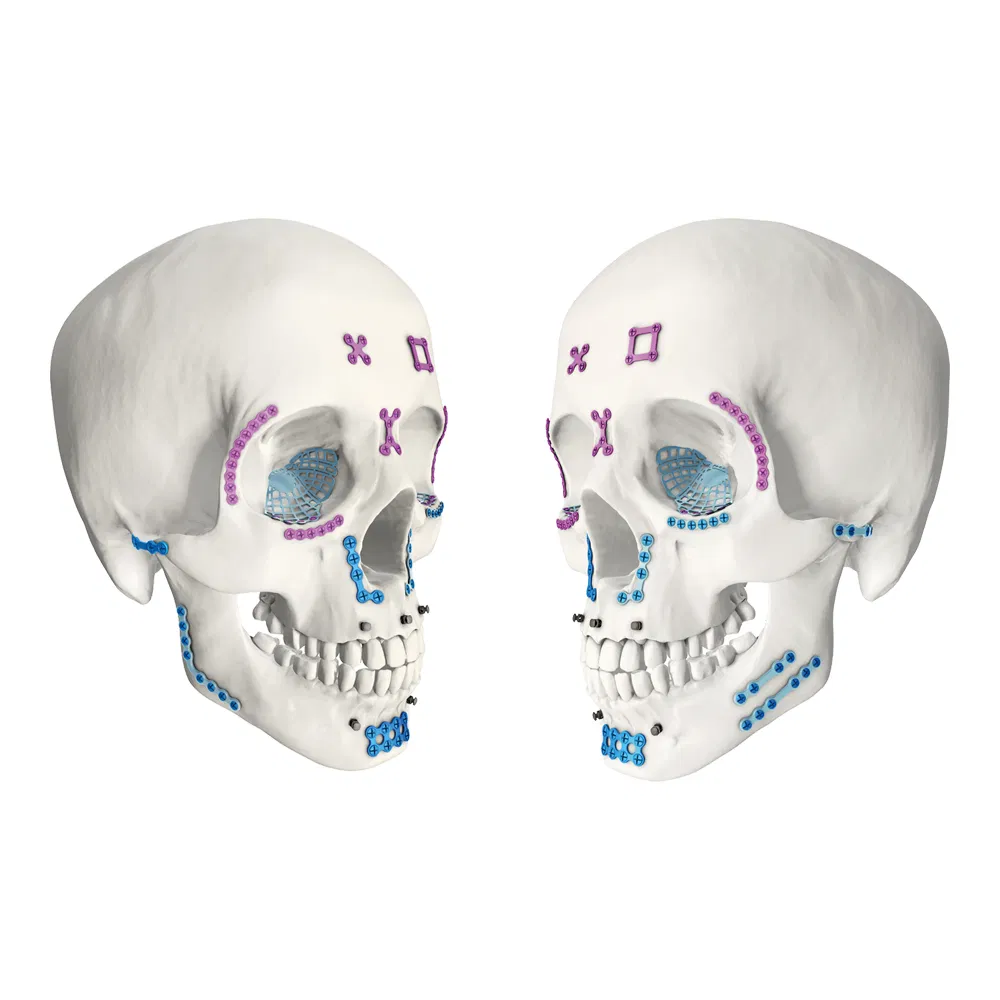
| Maxillofacial Micro Locking X-Plate | 4 holes, 0.6mm, 14mm |
| Maxillofacial Micro Locking Rectangular Plate | 4 holes, 0.6mm, 14*14mm |
| Maxillofacial Micro Locking Straight Bridge Plate | 4 holes, 0.6mm, 20mm |
| Maxillofacial Micro Locking Straight Plate | 6-14 holes, 0.6mm, 24-56mm |
| Maxillofacial Micro Locking Arc Plate | 6-14 holes, 0.6mm, 24-49mm |
| Maxillofacial Micro Locking 110° L-plate | 4-5 holes, 0.6mm, 16-20mm |
| Maxillofacial Micro Locking 90° L-plate | 4 holes, 0.6mm, 16mm |
| Maxillofacial Micro Locking Y-plate | 6 holes, 0.6mm, 17mm |
| Maxillofacial Micro Locking T-plate | 5-7 holes, 0.6mm, 16-24mm |
*All the prices are only for reference. Exact price will be based on the latest quotation.




 Voila Orthopedic Implants
Voila Orthopedic Implants