














| Customization: | Available |
|---|---|
| Type: | Surgical Supplies Materials |
| Material: | Peek |

Bullet Lumbar Spine System products are used for:
Clinical Versatility: Suitable for multiple surgical approaches including posterior (PLIF, TLIF) and anterior (ALIF) methods.
Our Bullet Lumbar Spine System provides an effective solution for lumbar fusion surgery with its precision manufacturing, diverse size options, modern design, and enhanced fixation capabilities. Features include large-size cutting threads, thread design for stability, and high flexibility for rapid fusion.



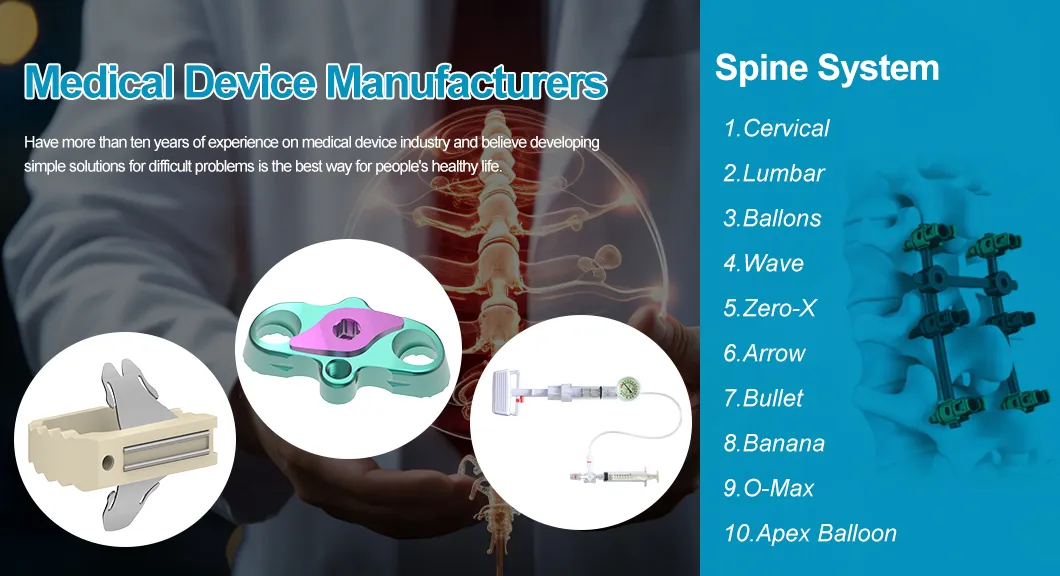
Our company was established in 2008 by pioneers of the modern orthopedic implant industry. With over ten years of experience in the medical device sector, we believe in developing simple solutions for complex clinical challenges to improve patient health.
We are devoted to providing innovative products, including orthopedic implants, surgical staplers, and wound care systems. We aim to support medical professionals worldwide with high-quality instruments, working side-by-side with surgeons to achieve maximum patient well-being.


 Voila Orthopedic Implants
Voila Orthopedic Implants