


1 / 5










| Still deciding? Get samples of $ ! US$ 1600/Set Request Sample |
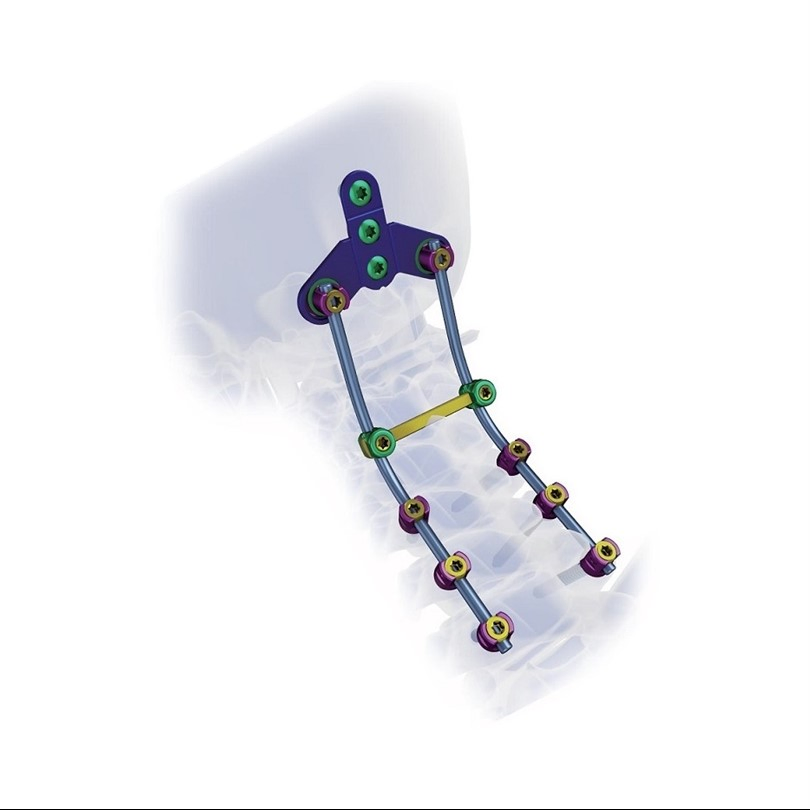
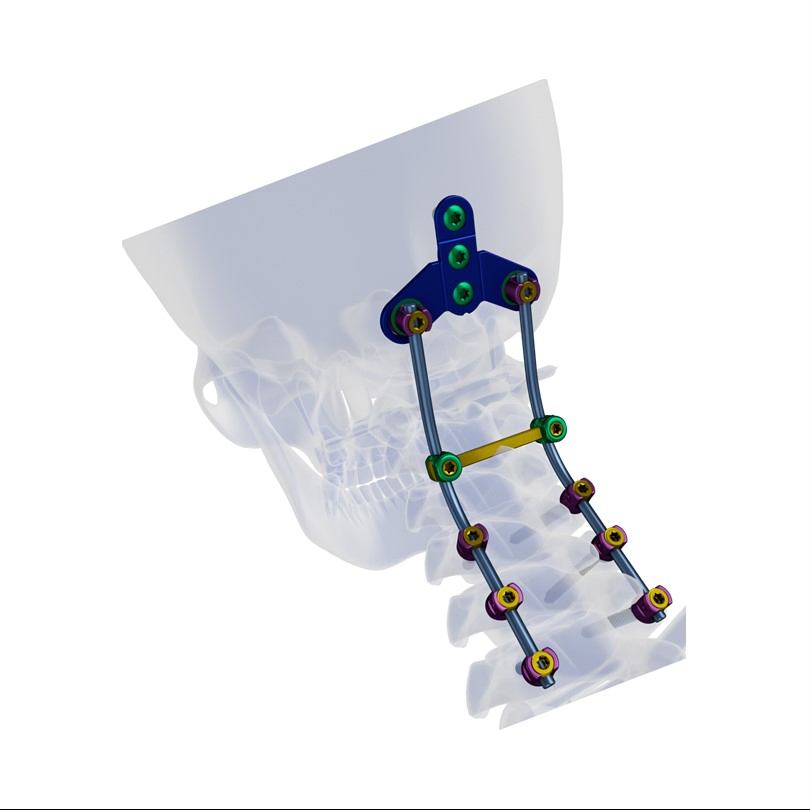
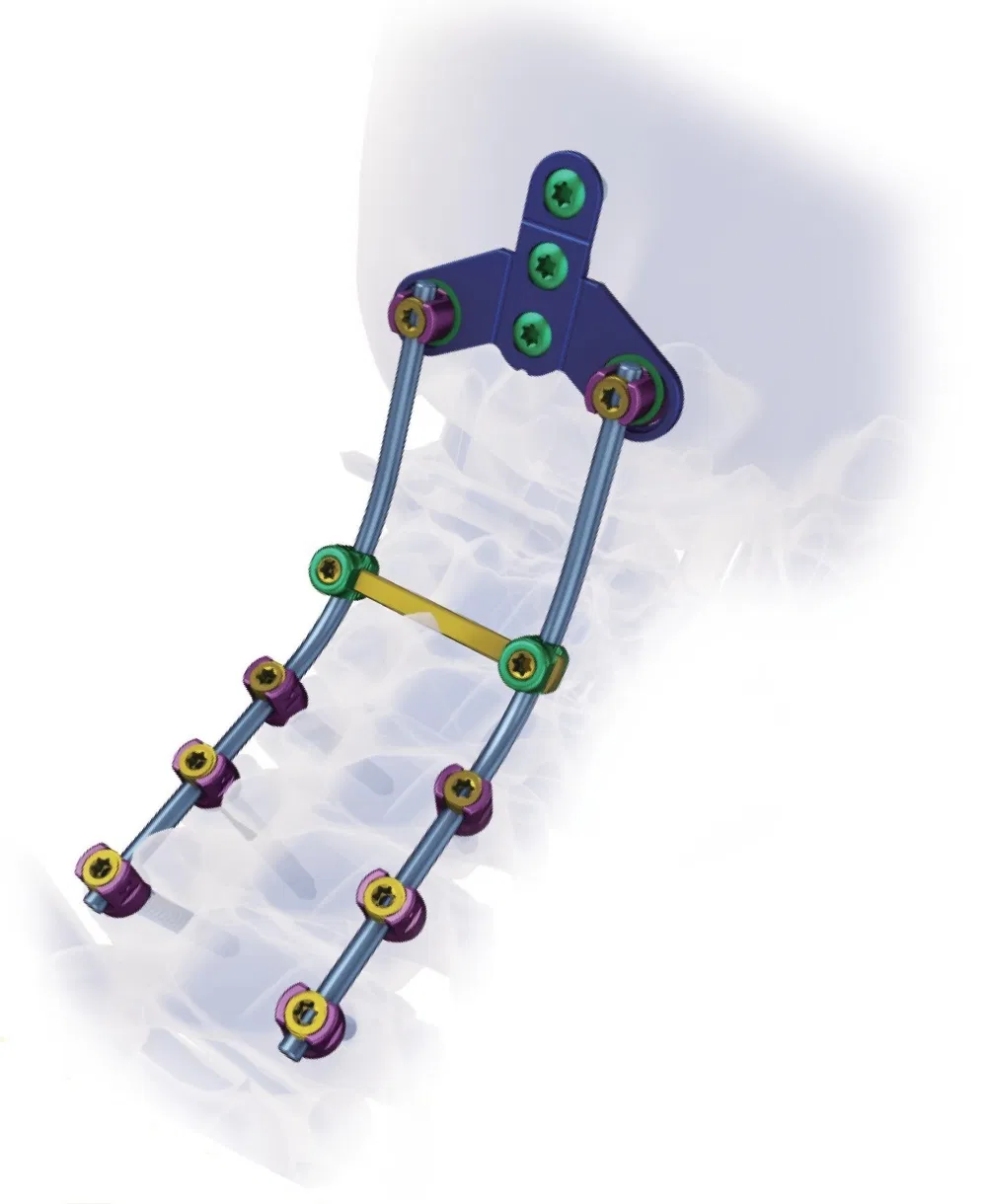
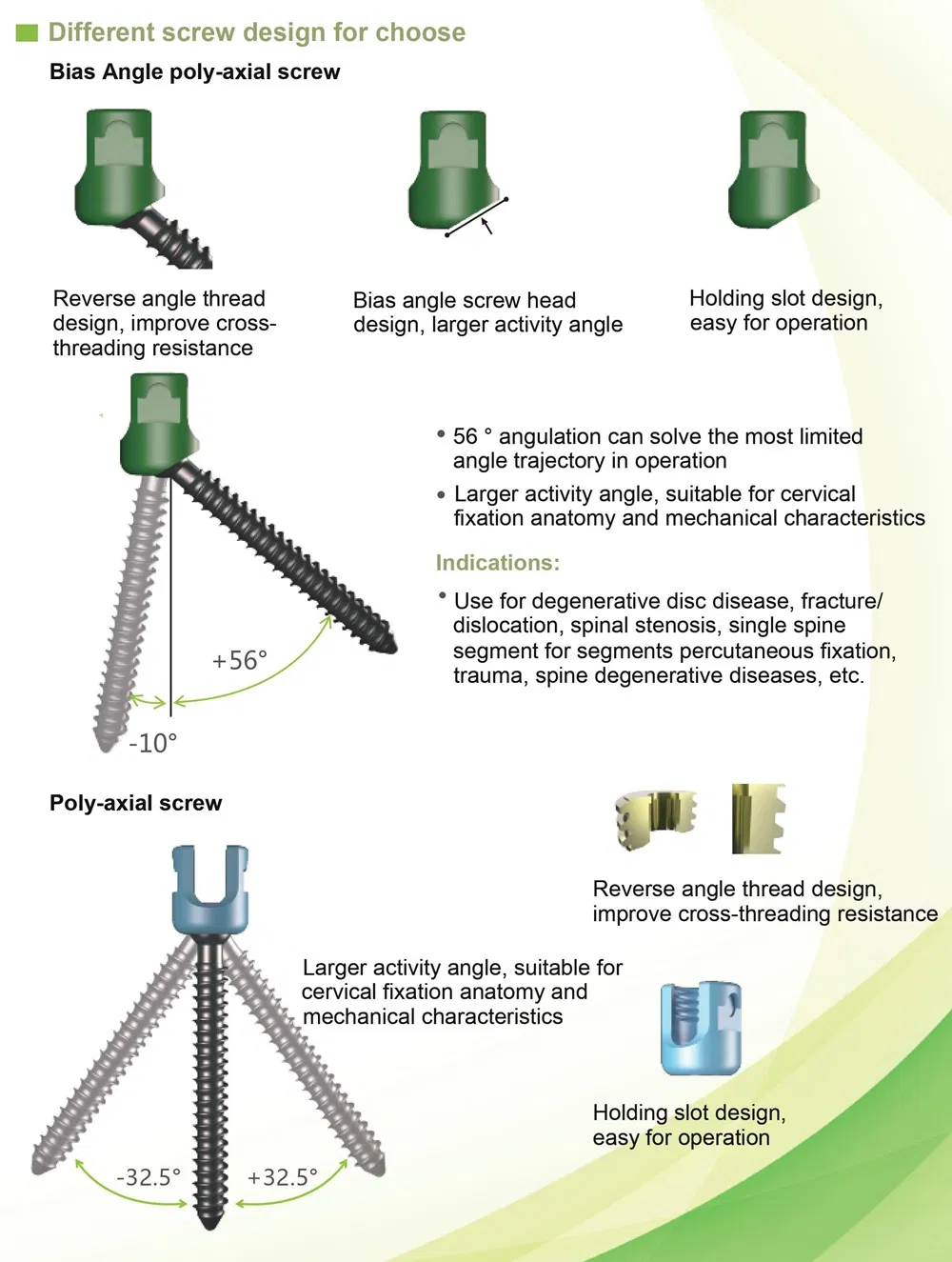
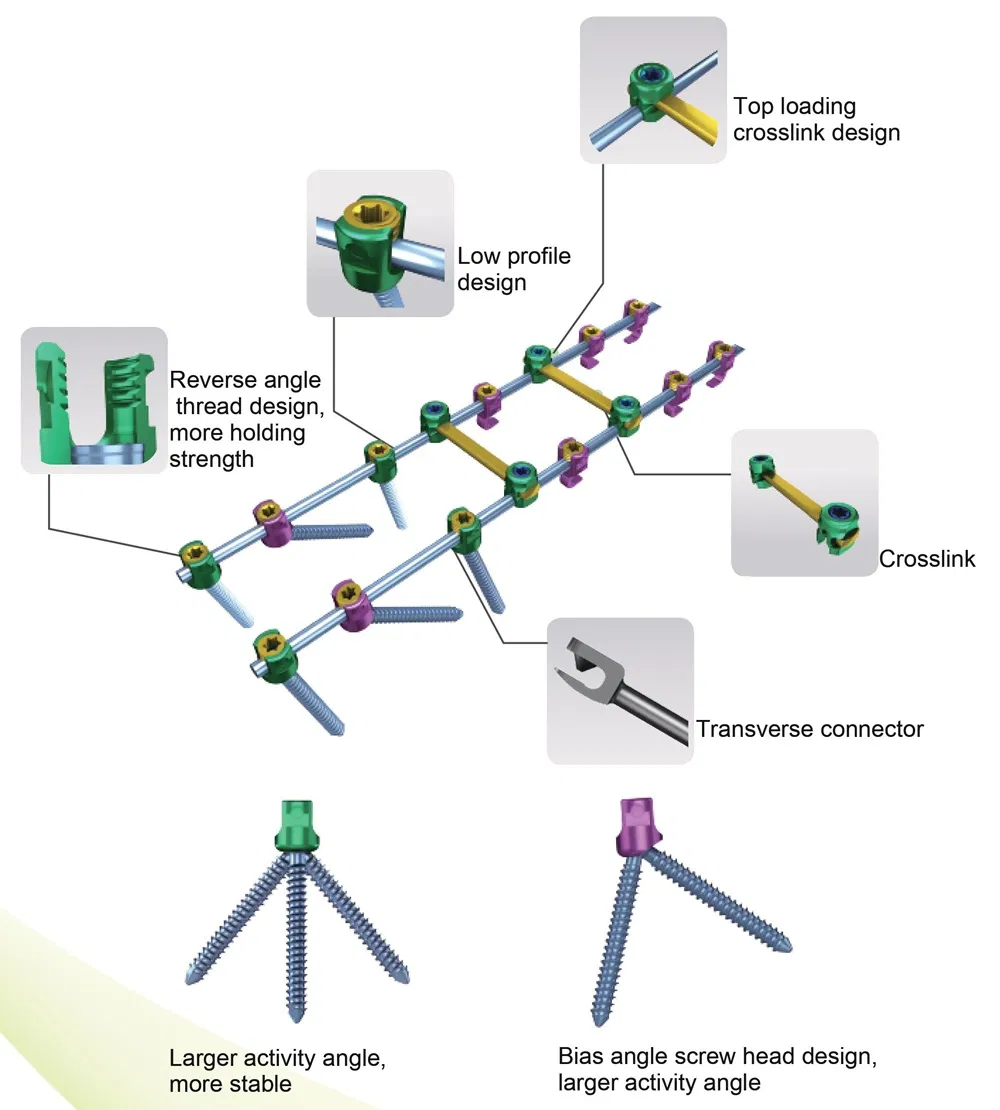
Creation 3.5 Posterior Cervical Fixation System (OTC connect)
This advanced orthopedic spinal implant system is designed for posterior cervical surgery, providing stable fixation and ease of use for medical professionals.
Superior biocompatibility makes this system suitable for addressing various spinal conditions including:


Packing Details:
Delivery Methods:

Core Focus: Providing one-stop orthopedic product solutions since 2016. Our production capacity reaches 10,000 units per month, serving clients across Southeast Asia, the Middle East, Latin America, Africa, and Europe.
Factory Advantages:



 Voila Orthopedic Implants
Voila Orthopedic Implants