













| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |
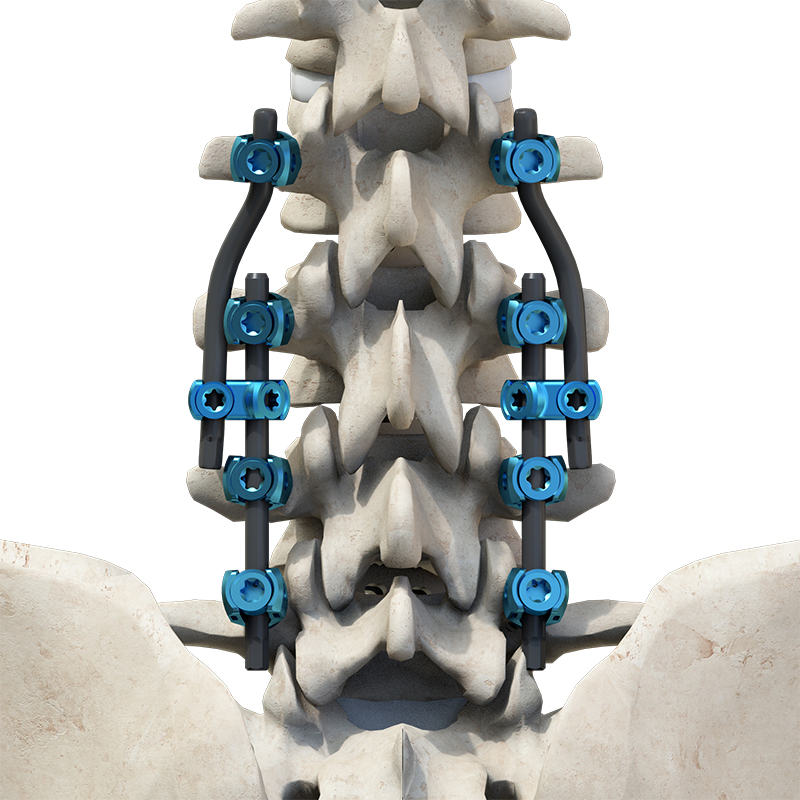
The Thoracolumbar Posterior Pedicle Screw and Rod System consists of high-quality components including pedicle screws, rods, set screws, crosslinks, lateral connectors, and hooks. This system is engineered to restore spinal alignment, correct deformities, and provide both immediate and long-term biomechanical stability following posterior thoracolumbar procedures.
By placing pedicle screws through the vertebral pedicles and connecting them with contoured rods, the system facilitates essential surgical maneuvers such as distraction, compression, derotation, and realignment. The resulting stable three-dimensional construct supports load-sharing and promotes successful spinal fusion.
Material Composition: Components are manufactured from TC4 ELI titanium alloy, CoCrMo alloy (Co28Cr6Mo), and pure titanium (TA2G) for optimal performance.


| Model Number | Specification (mm) |
|---|---|
| 1072401 | S |
| M |

Professional Orthopedic Manufacturer You Can Trust
As a leading manufacturer of standardized orthopedic implants and instruments, we serve global distributors with a full range of products covering trauma, spine, joints, sports medicine, and CMF.
Our facility features in-house R&D, a certified laboratory, and modern production lines. We focus on reliable, scalable manufacturing with low MOQ and fast turnaround to support high-volume distribution and private label needs.
 Voila Orthopedic Implants
Voila Orthopedic Implants