
1 / 5










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
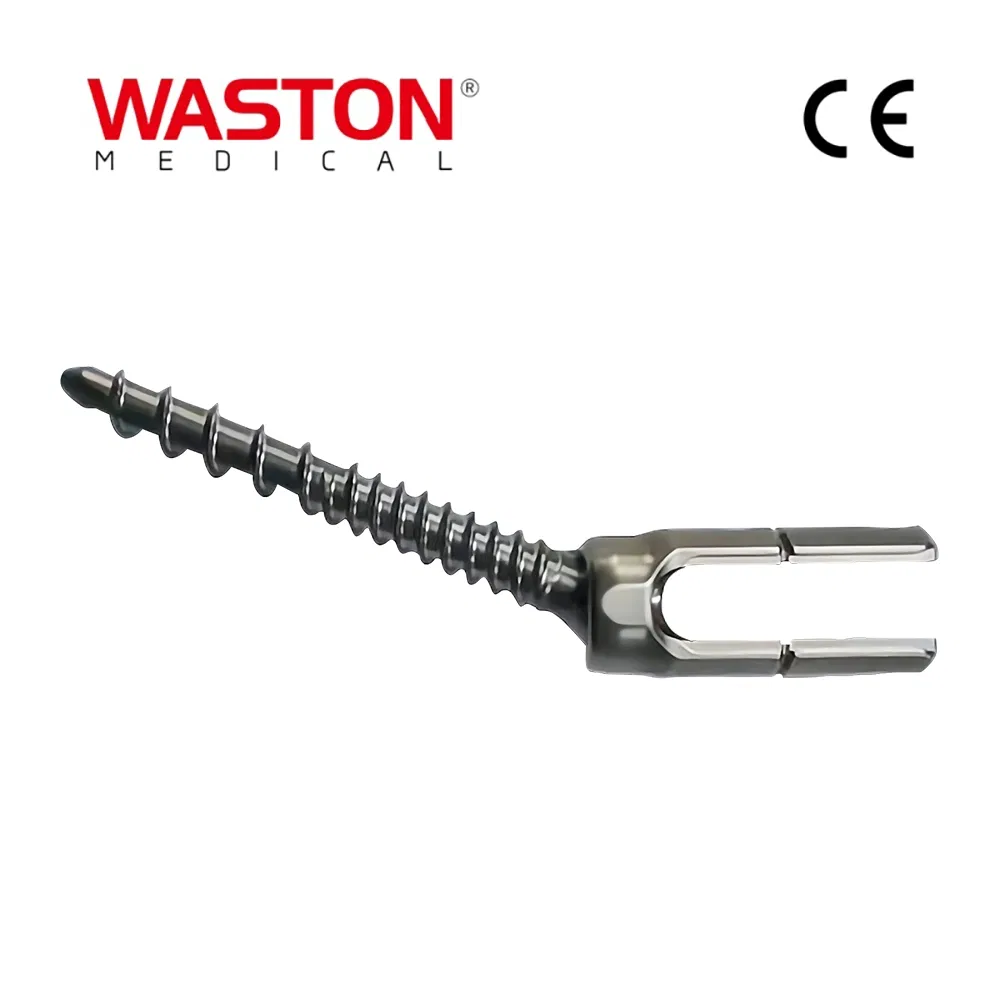
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 |

Our commitment to R&D investment exceeds 8% of total sales annually, transforming clinical experience into patented medical innovations. We utilize advanced German, American, and Japanese-imported machining centers to ensure high-precision production of spinal systems and orthopaedic implants.


Operating strictly under ISO13485 and CE MDD 93/42/EEC directives, our testing center includes biomechanical laboratories and physical-chemical testing centers to ensure raw material control and product performance analysis.
Our products are exported globally, meeting international standards across various continents. We serve as a major OEM supplier for global leaders in the orthopaedic industry.



 Voila Orthopedic Implants
Voila Orthopedic Implants