


1 / 3






| Still deciding? Get samples of $ ! US$ 85/Piece Request Sample |
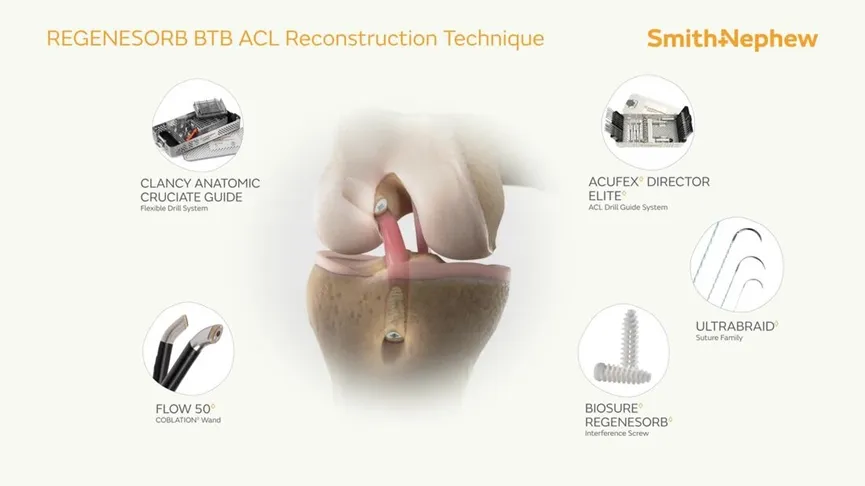
The Interface Screw is designed for use in ACL reconstruction surgeries to secure tendon or ligament grafts in the femoral and tibial bone tunnels. It provides a strong and reliable fixation by compressing the tendon or ligament graft directly against the bone tunnel wall. The screw's unique mechanism ensures optimal stability and a firm attachment between the soft tissue graft and the bone.


 Voila Orthopedic Implants
Voila Orthopedic Implants