











| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |

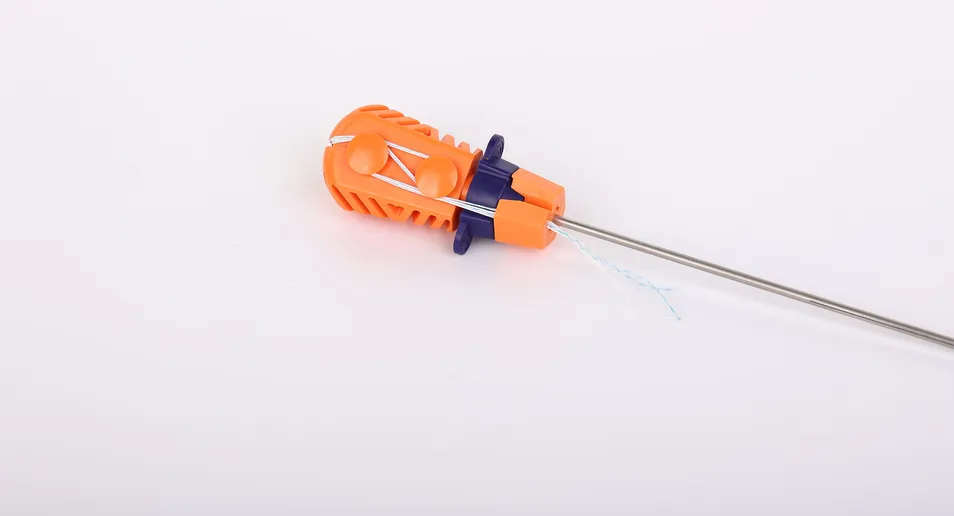
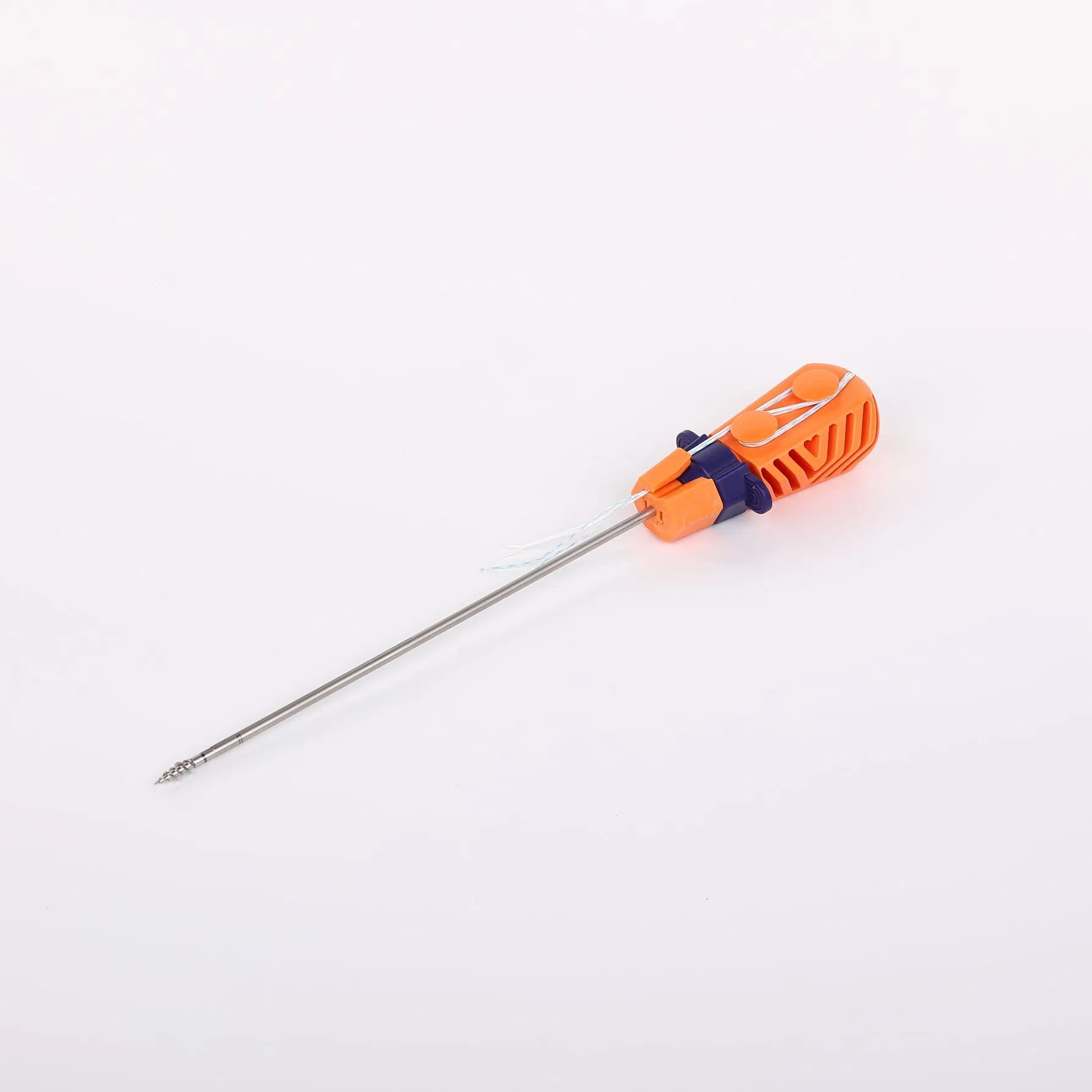
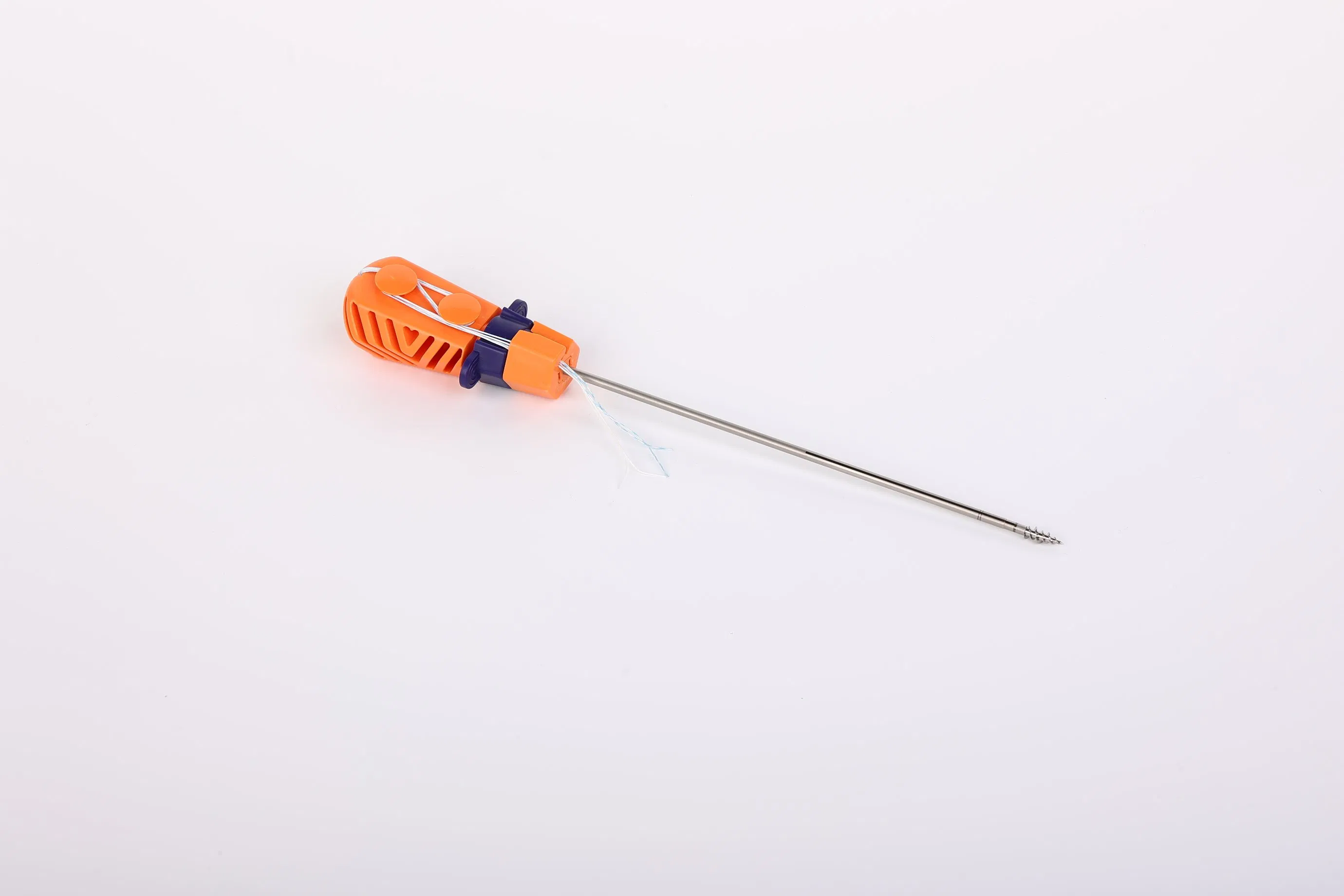
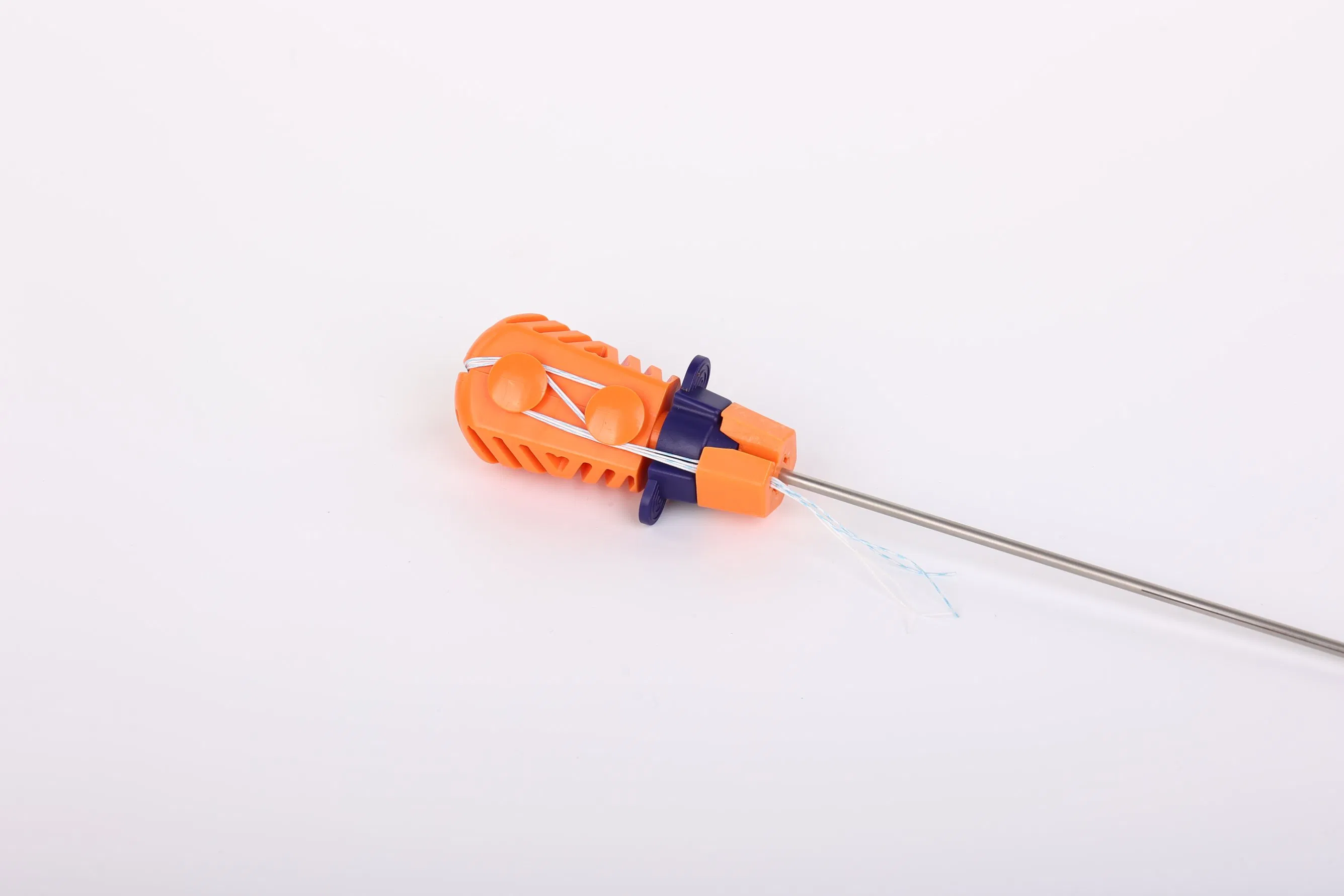
Suture Anchor System consists of anchor, nonabsorbable suture and inserter. The anchor is made of Ti6Al4V alloy, which meets the requirement of ISO5832-3:2016. The suture is made of ultra high molecular weight polyethylene (UHMWPE) material without coating, woven from several UHMWPE yarns (ASTM F2848-17).
The blue suture is made from white UHMWPE dyed with medical-grade blue material (code 73.1015), approved by regulatory standards. The inserter parts contacting the human body are made of stainless steel (ASTM F899-12b), while the handle is constructed from durable polycarbonate and ABS material.


Our comprehensive range includes Suture Anchor Systems, Meniscal Repair Systems, and other orthopedic surgical consumables. With advanced production capabilities, these products have gained access to more than 40 countries across the globe.
To guarantee product quality and safety, investments have been made in a Class 100,000 clean workshop and a Class 10,000 purification laboratory, underpinning full-cycle capabilities in R&D and manufacturing. All products are fully compliant with CE certification and ISO 13485 standards.



 Voila Orthopedic Implants
Voila Orthopedic Implants