🔹 Basic Specifications
Certification
CE, ISO13485
Applicable Population
General Group
Function
Connect Soft Tissue and Bone
Material
Peek, Titanium Screw
Available Sizes
5.0mm to 6.5mm, 1.8mm to 4.5mm


📝 Product Description
⚙️
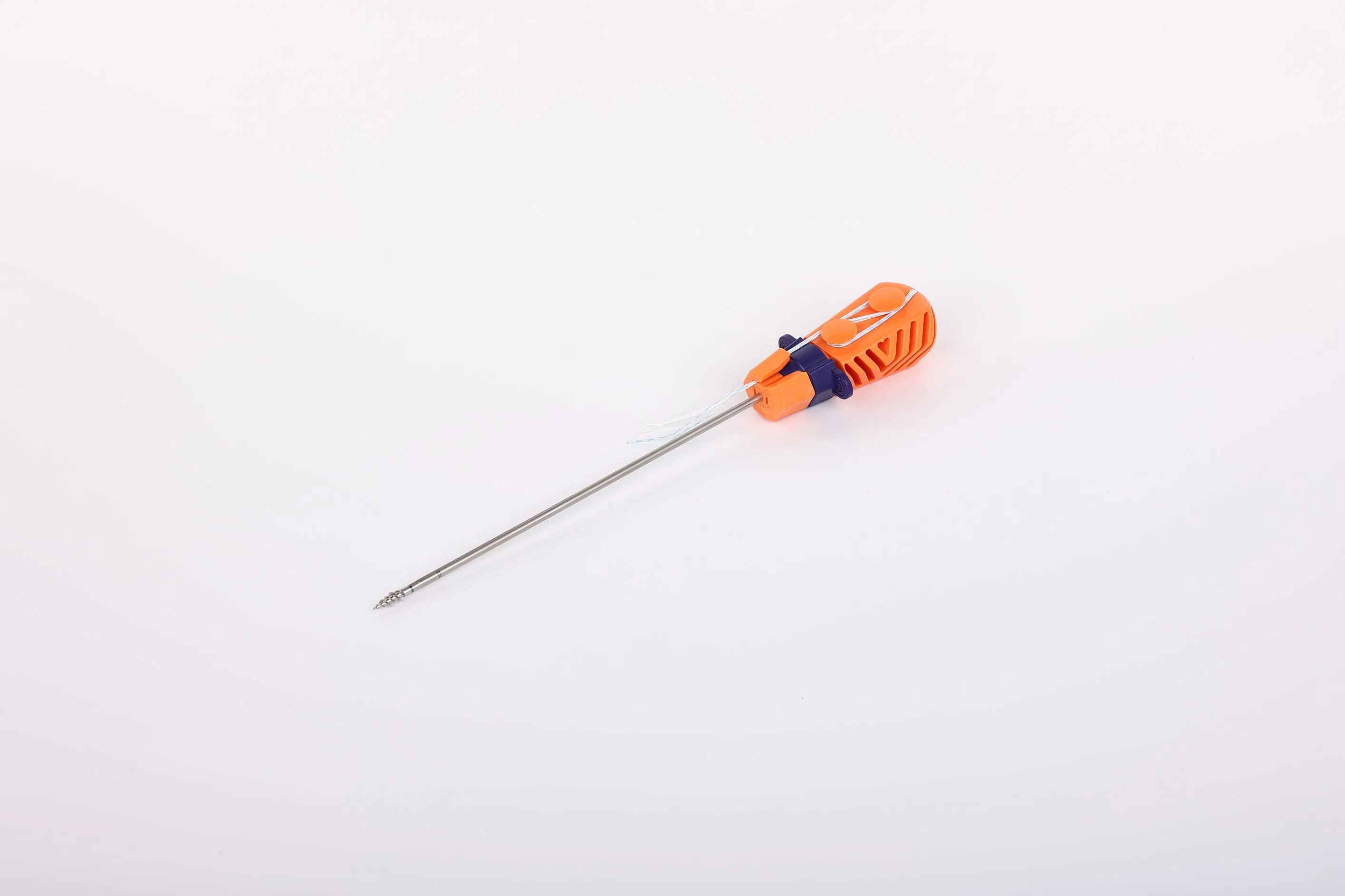
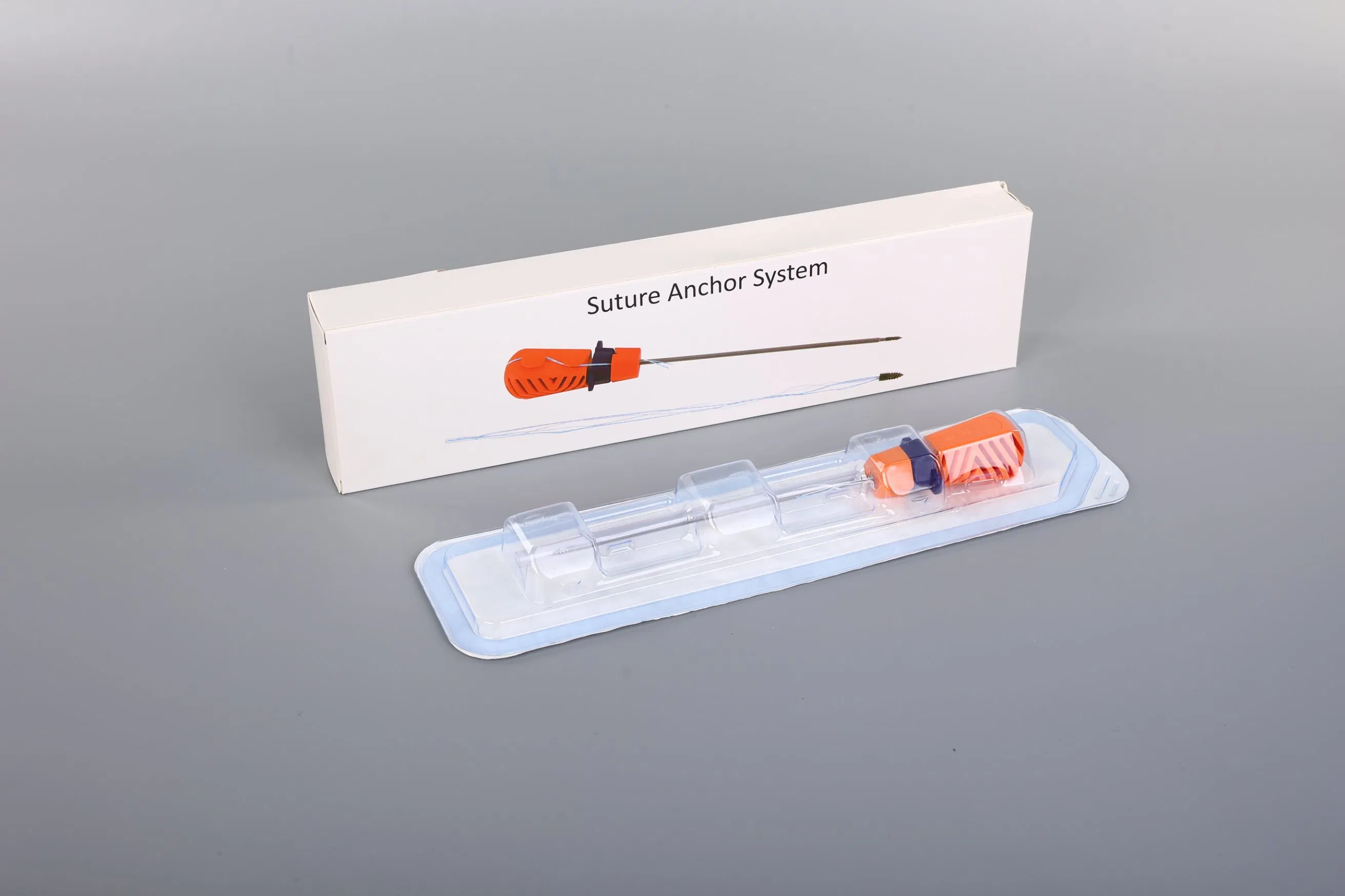
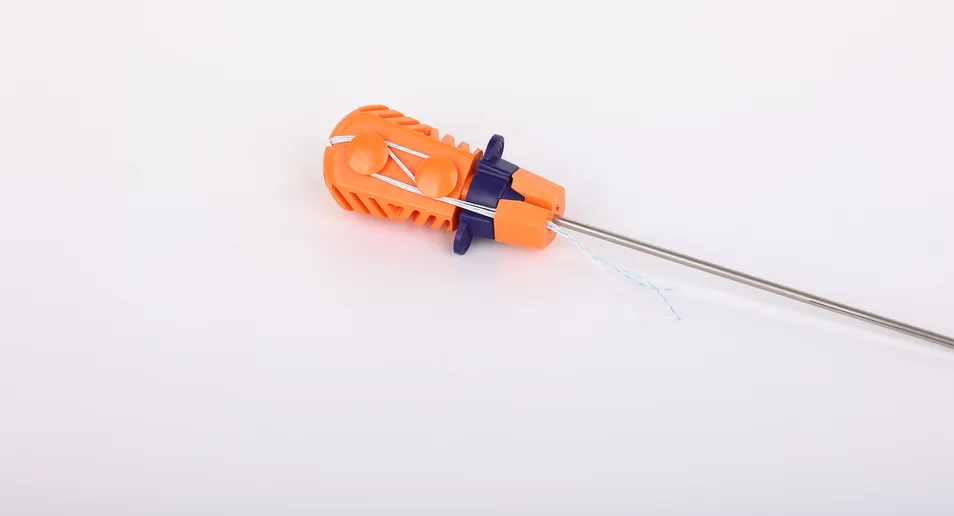
System Composition: The Suture Anchor System consists of an anchor, non-absorbable suture, and an inserter.
Anchor Material: The anchor is manufactured from Ti6Al4V alloy, strictly meeting ISO5832-3:2016 requirements for medical implants.
Suture Properties: High-performance sutures are made of Ultra-High Molecular Weight Polyethylene (UHMWPE) without coating, woven from several UHMWPE yarns (ASTM F2848-17 compliant). The blue sutures are colored with FDA-approved materials.
Inserter Design: The parts contacting the human body are crafted from stainless steel (ASTM F899-12b). The ergonomic handle is made of medical-grade polycarbonate and ABS material.

❓ Frequently Asked Questions
Q: What materials are used for the anchors?
A: The anchors are primarily made of Ti6Al4V Titanium alloy or PEEK material, ensuring biocompatibility and strength during joint repair surgery.
Q: Is the suture absorbable or non-absorbable?
A: The system uses high-strength, non-absorbable sutures made of Ultra-High Molecular Weight Polyethylene (UHMWPE).
Q: Are the products certified for international use?
A: Yes, the suture anchor system is fully compliant with CE certification and ISO 13485 standards.
Q: Do you offer different sizes for specific surgical needs?
A: Yes, anchors are available in various diameters including 1.8mm, 2.8mm, 3.5mm, 4.5mm, 5.0mm, 5.5mm, and 6.5mm to suit different orthopedic procedures.
Q: Is the system provided sterile?
A: Yes, all surgical components are provided EO Sterile and are intended for single use to ensure maximum safety.
Q: Can I request customized OEM services?
A: Yes, we provide comprehensive OEM services including custom design and specialized packaging solutions.




















 Voila Orthopedic Implants
Voila Orthopedic Implants