Trusted by surgeons and distributors across 80+ countries. Manufactured under ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP compliance standards.
We are one of the most reputed manufacturers and exporters of Orthopedic Implants and Instruments — a Star Export House company certified by the Government of India, proudly serving the global medical community.
We are exporting to more than 80 countries worldwide and pride ourselves on Superior Quality and Competitive Pricing. We operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe and USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price. We believe human life is invaluable. We understand your requirements, we recognize your needs — come partner with us.
Our manufacturing facilities are equipped with state-of-the-art CNC machining centers, precision grinding machines, and advanced surface treatment lines. Every product undergoes rigorous dimensional inspection, material testing, and sterility validation before it leaves our facility.
We are considered as a top Trauma & Orthopedic Implants company, trusted by orthopedic surgeons, hospitals, and distributors across Europe, USA, Latin America, the Middle East, Africa, and Asia.
Our Commitment to You:
"We don't compromise on quality in exchange for a cheaper price — because human life is invaluable. Every implant we manufacture carries the weight of our responsibility to patients worldwide.
From raw material sourcing to final packaging, every step is monitored, tested, and validated to ensure that what reaches the surgeon's hand is nothing short of perfect."
Our modern production plant combines precision engineering with strict quality protocols — designed to meet the highest international medical device manufacturing standards.

CNC precision machining & quality production lines

Stringent testing for international compliance
We have a highly equipped R&D lab and stringent quality control standards to ensure our products meet international benchmarks at every stage of production.
Quality Control starts from the raw material stage, enabling us to control quality of every component that goes into manufacture of the finished product. We use only certified medical-grade alloys and biomaterials.
Every production batch undergoes multiple in-process inspection points using CMM machines, surface profilometers, and hardness testers to ensure dimensional accuracy and surface integrity.
Before dispatch, all products are subjected to final inspection, sterility testing, and documentation review to ensure full compliance with CE, ISO 13485, and WHO-GMP requirements.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from customers to create new designs meeting the future needs of Orthopedic Surgeons worldwide.
We comply with ISO 9001:2015, EN ISO 13485:2016, CE marking, and WHO-GMP guidelines — ensuring our products are accepted by regulatory bodies across all major global markets.
Complete lot traceability from raw material to finished product is maintained. Our documentation system ensures rapid response to any regulatory or customer query, supporting post-market surveillance.
What sets us apart from other orthopedic implant manufacturers in the global market — built on decades of experience and continuous improvement.
Active export operations to 80+ countries with established logistics partnerships and customs expertise for seamless delivery worldwide.
Direct manufacturer pricing without intermediaries. We offer the best value without ever compromising on product quality or patient safety.
From trauma implants to spine systems, joint replacement, and surgical instruments — a comprehensive one-stop solution for orthopedic needs.
Efficient production planning and large inventory ensure quick order fulfillment. Standard orders dispatched within 7–15 business days.
Our quality management system is validated by internationally recognized certification bodies, providing our global partners with full regulatory confidence.
International standard for Quality Management Systems — ensuring consistent product quality and continuous improvement across all processes.
Medical device-specific quality management standard — the gold standard for orthopedic implant manufacturers supplying to European and global markets.
CE certification confirms our products meet EU safety, health, and environmental requirements — mandatory for sales in the European Economic Area.
Good Manufacturing Practice compliance as per WHO guidelines ensures our manufacturing environment, processes, and documentation meet global pharmaceutical-grade standards.
We offer comprehensive OEM and private label manufacturing services for orthopedic companies, distributors, and healthcare brands worldwide — from design to delivery.
Our R&D team works closely with your engineers to develop custom implant designs, modifications, or entirely new product lines tailored to your market's surgical requirements.
Rapid prototyping using CNC machining followed by full mechanical and biocompatibility testing. We provide complete design validation documentation for regulatory submissions.
Full private labeling service with your brand identity, custom packaging, sterilization, and labeling compliant with EU MDR, FDA, and other regional regulations.
We provide complete technical files, Declaration of Conformity, and product dossiers to support your CE marking, FDA 510(k), and other country-specific registration processes.
From small trial orders to high-volume production runs — our flexible manufacturing capacity scales with your business growth without compromising quality or lead times.
Post-delivery support including complaint management, product traceability records, and field safety corrective action support to help you maintain regulatory compliance.
A complete portfolio of orthopedic implants and surgical instruments designed to support orthopedic surgeons in trauma, spine, joint reconstruction, and sports medicine.
Locking plates, intramedullary nails, screws, and external fixators for fracture management.
Total hip, knee, and shoulder arthroplasty systems engineered for longevity and patient outcomes.
Pedicle screw systems, interbody cages, cervical plates, and dynamic stabilization devices.
A full range of orthopedic surgical instruments — reamers, drill bits, retractors, and custom instrument sets.
Trusted by leading distributors, hospitals, and orthopedic companies across Europe, the USA, and beyond — here's what our partners say about working with us.
"We have been sourcing orthopedic implants from this company for over 8 years. The quality is consistently excellent, and their ISO 13485 documentation makes our EU regulatory submissions seamless. Highly recommended."
"Their OEM capability is outstanding. We approached them with a custom implant design and they delivered prototypes within 3 weeks, with full validation documentation. Their R&D team is truly professional."
"Competitive pricing without quality compromise — that's rare in this industry. We've tested their trauma plates against European brands and the dimensional accuracy is identical. Great partner for our hospital procurement."
We combine manufacturing excellence, regulatory expertise, and a genuine commitment to patient safety — making us the preferred sourcing partner for orthopedic companies worldwide.
As a direct manufacturer, we eliminate middlemen — giving you better pricing, faster communication, and full control over product specifications and customization.
Unlike many competitors, we never trade quality for price. Our zero-compromise quality policy is backed by multi-stage inspection and certified quality management systems.
With 80+ countries served and repeat customers from Europe and the USA, our track record speaks for itself. We are a trusted name in global orthopedic supply chains.
From product development and manufacturing to packaging, labeling, sterilization, and logistics — we handle the entire supply chain so you can focus on your business.
We invest in understanding your business, your market, and your patients. Our team provides dedicated account management and proactive support for long-term success.
Our R&D team consistently develops new products and improved designs, ensuring our partners always have access to the latest advancements in orthopedic implant technology.
Join 80+ countries that trust us for superior quality orthopedic implants and instruments. Let's discuss your requirements today.
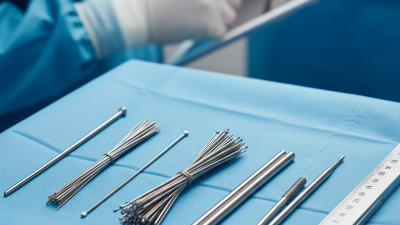
When it comes to selecting medical instruments, choosing the right Kirschner Wire is essential for successful procedures. These wires are often used
Author: Mason Date: 2026-03-30
Learn More
In the realm of orthopedic surgery, Titanium Spinal Mesh stands out as a revolutionary material. Dr. Emily Zhang, a leading expert in spinal
Author: Liam Date: 2026-03-27
Learn More
The 2026 Canton Fair is set to showcase exciting innovations in the medical device sector, including the latest trends in Cortex Bone Screws. With a
Author: Isabella Date: 2026-03-25
Learn More