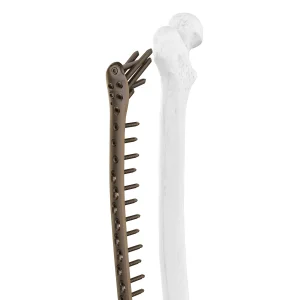
 Canwell Proximal Femoral Locking Plate Femur Plate Large Fragment Femoral Plate
Canwell Proximal Femoral Locking Plate Femur Plate Large Fragment Femoral Plate
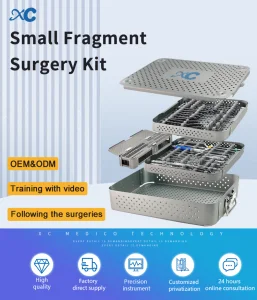 Orthopedic Surgical Trauma Implant Distal Radius and Proximal Humerus Locking Plate Instrument Small Fragment Set
Orthopedic Surgical Trauma Implant Distal Radius and Proximal Humerus Locking Plate Instrument Small Fragment Set
 Tibial Intramedullary Nail Instrument Set for Orthopedic Tibia Interlocking Nail Instruments
Tibial Intramedullary Nail Instrument Set for Orthopedic Tibia Interlocking Nail Instruments
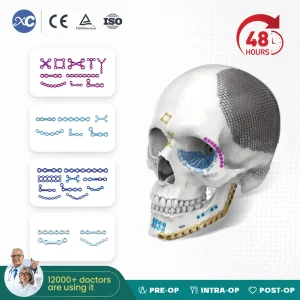 Cmf System Titanium Material Orthopedic Surgery Maxillofacial Locking Plates and Screws Implant
Cmf System Titanium Material Orthopedic Surgery Maxillofacial Locking Plates and Screws Implant
 Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
 Czmeditech Orthopedic Tibia Intramedullary Nail
Czmeditech Orthopedic Tibia Intramedullary Nail
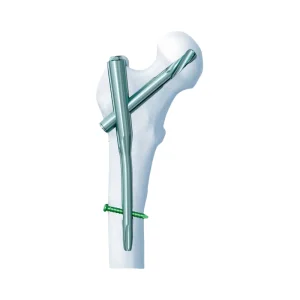 Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral Intramedullary Implants Pfna Proximal Femur Interlocking Nail
Gama Medical Surgical Bone System Pfn Titanium Orthopedic Trauma Surgery Femoral Intramedullary Implants Pfna Proximal Femur Interlocking Nail
 Orthopedic Plates Titanium Medical Surgical Fracture Bone Trauma Large Fragment Proximal Femur Neck Plate with Hook Locking Plate with CE
Orthopedic Plates Titanium Medical Surgical Fracture Bone Trauma Large Fragment Proximal Femur Neck Plate with Hook Locking Plate with CE
The global orthopedic implant market is witnessing a transformative era, driven by the increasing incidence of musculoskeletal disorders and high-energy trauma cases. The Proximal Tibia Locking Plate has emerged as a gold standard in the fixation of complex plateau fractures. As population demographics shift toward an aging society, the demand for osteoporotic fracture management has skyrocketed, placing China at the epicenter of medical device innovation and supply.
Today, the Proximal Tibia Locking Plate manufacturer sector is no longer just about basic mechanical fixation. It is about biomechanical compatibility, anatomical contouring, and minimally invasive surgical (MIS) access. From the bustling surgical theaters in New York to the developing medical centers in Southeast Asia, these plates are essential for restoring patient mobility and reducing post-operative complications like malunion or hardware failure.
Modern surgery requires more than just a "one-size-fits-all" approach. As a premier Proximal Tibia Locking Plate supplier, we focus on anatomical pre-contouring. This eliminates the need for intraoperative bending, preserving the structural integrity of the titanium alloy and reducing surgery time.
Trends indicate a shift toward Variable Angle (VA) Locking Technology. This allows surgeons to direct screws into specific bone fragments, ensuring maximum stability in comminuted fractures. Our R&D team utilizes AI-driven simulation to test load-bearing capacities, ensuring our plates withstand the rigorous demands of early weight-bearing rehabilitation.
Whether it is the lateral proximal tibia or the medial plateau, our locking systems provide the necessary "internal-external fixator" effect, bridging the fracture zone while preserving the periosteal blood supply.
We utilize Grade 5 Titanium (Ti-6Al-4V) ELI, offering superior biocompatibility and fatigue resistance compared to traditional stainless steel. Every batch undergoes ultrasonic testing.
In an era of global logistics uncertainty, our massive manufacturing capacity in China ensures a consistent supply of Proximal Tibia Locking Plates for government tenders and private hospitals.
By optimizing production through CNC automation and AI quality inspection, we offer competitive pricing without compromising the surgical outcomes or material purity.
Procurement officers and surgical distributors globally are seeking partners who provide not just a product, but a full system. This includes specialized Orthopedic Trauma Instrument Sets, tailored for smooth plate insertion and secure screw locking. Our integrated approach helps hospitals reduce the total cost of ownership while maintaining a high standard of care.
From the high-impact sports injuries seen in European ski resorts to the road traffic accidents prevalent in emerging economies, the Proximal Tibia Locking Plate is a versatile tool. In geriatric patients, the locking mechanism provides superior purchase in osteoporotic bone, preventing the "settling" of the articular surface.
Our plates are designed for MIPPO (Minimally Invasive Percutaneous Plate Osteosynthesis) techniques. This approach minimizes soft tissue disruption, which is critical in the proximal tibia where the skin cover is thin and the risk of wound complications is high.
As a leading exporter, we provide comprehensive surgical guides and technical training to ensure that surgeons in every corner of the world can achieve the best possible patient outcomes.
We are most reputed manufacturers/exporters of Orthopedic Implants and Instruments & Star Export House company certified by Government.
We are exporting to more than 80 countries worldwide & pride ourselves on Superior Quality and Competitive Pricing, and we operate under a quality system that complies with ISO 9001:2015, EN ISO 13485:2016, CE & WHO: GMP Compliance. Many reputed companies in Europe and USA regularly buy from us.
Unlike many of our competitors, we don't compromise on quality in exchange for a cheaper price !! We believe human life is invaluable !! We understand your requirements, we recognize your needs! Come partner with us...


We have a highly equipped R&D lab and stringent quality control standards to ensure quality of our products as per international standards. Quality Control at Narang Medical starts from the raw material stage. This makes it possible for us to control the quality of every component that goes into manufacture of the finished product.
Our R&D team consists of highly qualified engineers who persistently conduct research and constantly seek feedback from the customers as they create new designs to meet the future needs of Orthopedic Surgeons worldwide. We are thus considered as a top Trauma & Orthopedic Implants company.
 Orthopedic PFNA Femoral Implant PFN Proximal Femur Intramedullary Interlocking Nail
Orthopedic PFNA Femoral Implant PFN Proximal Femur Intramedullary Interlocking Nail
 Intramedullary Nail System Expert Humeral Proximal Interlocking Nail Blade Locking
Intramedullary Nail System Expert Humeral Proximal Interlocking Nail Blade Locking
 Ao Design Orthopedic Surgical Instrument Ring Ilizarov Tibia Femur Lower Limb Lengthening External Fixator Price Fixation for Femur Tibia Fracture Surgery
Ao Design Orthopedic Surgical Instrument Ring Ilizarov Tibia Femur Lower Limb Lengthening External Fixator Price Fixation for Femur Tibia Fracture Surgery
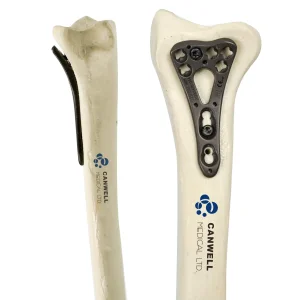 Canwell Variable Angle Radius Locking Plate Orthopaedic Implants Titanium Locking Plate
Canwell Variable Angle Radius Locking Plate Orthopaedic Implants Titanium Locking Plate
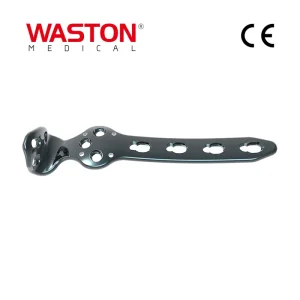 Quality Assurance Titanium Trauma Ankle Locking Plate Bone Screw Instrument Set Used for Tibia&Fibula Orthopedic Surgical Implant
Quality Assurance Titanium Trauma Ankle Locking Plate Bone Screw Instrument Set Used for Tibia&Fibula Orthopedic Surgical Implant
 Excellent Quality Instrument Orthopedic Tibia & Percone External Fixator Orthopedic Surgical Instruments
Excellent Quality Instrument Orthopedic Tibia & Percone External Fixator Orthopedic Surgical Instruments
 Variable Angle Orthopedic LCP Distal Femoral Locking Plate
Variable Angle Orthopedic LCP Distal Femoral Locking Plate
 Proximal Lateral Femoral Plate -Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
Proximal Lateral Femoral Plate -Orthopedic Implant System for Femoral Shaft or Condylar Fracture Surgery
As the medical device industry evolves, sustainability and precision become paramount. Our factory in China is integrating Green Manufacturing processes to reduce waste in titanium production. Furthermore, we are exploring the integration of 3D-printed patient-specific implants for complex proximal tibia reconstructions where standard plates may not suffice.
Global distributors looking for a China Proximal Tibia Locking Plate manufacturer can rely on our advanced testing facilities. Every implant is subjected to dynamic fatigue testing, ensuring it exceeds the ASTM and ISO standards for load-bearing orthopedic devices. Our commitment to R&D ensures that we stay ahead of the curve, providing surgeons with the tools they need to improve patient lives.
Partnering with us means more than just sourcing products; it means gaining a long-term ally in the quest for medical excellence. Our global footprint across 80 countries is a testament to our reliability, quality, and vision for the future of orthopedic trauma surgery.