




1 / 5










| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
Indication: Low limbs fracture and lengthening.
Description: Used for fracture relevant to articular surface, with 4.8mm drill bit and used with olive-head kirschner wire if needed.
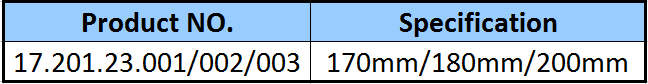
Description of Screws: 6.0/5.0 screw(SS/TA) is available.





 Voila Orthopedic Implants
Voila Orthopedic Implants