


1 / 5










| Customization: | Available |
|---|---|
| Application: | Orthopedic, Neurosurgery, Microsurgery |
| Material: | Steel |




| Component Description | Specification | Qty |
|---|---|---|
| Pin To Rod Coupling | φ8/4-5 | 12 |
| Rod To Rod Coupling | φ8/8 | 12 |
| 5-Hole Pin Clamp | φ8/5 | 3 |
| Straight Post | φ8 | 2 |
| 30° Post | φ8 | 4 |
| Self Drilling/Self Tapping Bone Screws | φ5*180 / 150 / 130 / 120 | 20 total |
| Carbon Fibre Rod | φ8*350 / 300 / 250 / 200 | 12 total |
| Instrument Set (Wrenches, Drill, Driver) | Various | 1 Set |
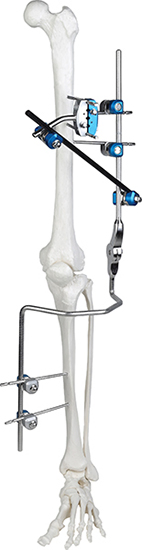
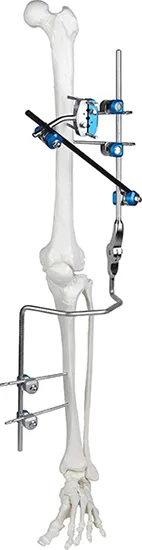
Knee Joint Fixator
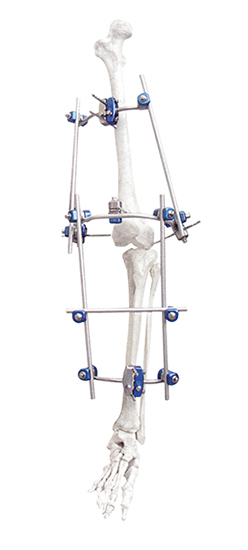
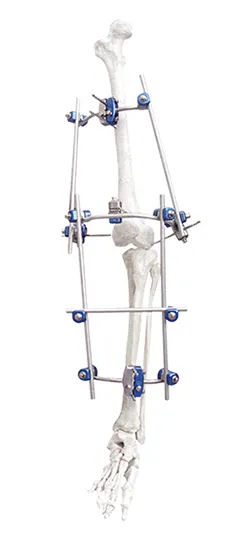
Knee Joint Mobilizer

Ankle Joint Fixator

Pelvic Fixator

Proximal Tibia External Fixator

Ankle Joint Mobilizer (Tibia Perone)
 Voila Orthopedic Implants
Voila Orthopedic Implants